Strategies for Preventing Microfluidic Channel Blockage in Single-Cell Analysis: From Fundamentals to Advanced Applications
Channel blockage presents a significant challenge in microfluidic single-cell analysis, impacting throughput, data reliability, and experimental efficiency.
Strategies for Preventing Microfluidic Channel Blockage in Single-Cell Analysis: From Fundamentals to Advanced Applications
Abstract
Channel blockage presents a significant challenge in microfluidic single-cell analysis, impacting throughput, data reliability, and experimental efficiency. This article provides a comprehensive examination of clogging mechanisms and prevention strategies across major microfluidic platforms, including droplet-based systems, valve-based chips, and hydrodynamic traps. By integrating foundational principles with practical methodological applications, troubleshooting protocols, and comparative validation frameworks, we deliver actionable insights for researchers and drug development professionals seeking to optimize system performance, maintain sample integrity, and ensure reproducible results in biomedical research and clinical applications.
Understanding Microfluidic Channel Blockage: Root Causes and Impact on Single-Cell Data Quality
Fundamental Clogging Mechanisms in Microscale Fluid Dynamics
This technical support center provides troubleshooting guides and FAQs to help researchers address and prevent clogging in microfluidic devices for single-cell analysis.
# FAQ: Understanding and Preventing Clogs
What are the primary physical mechanisms that cause clogging in microchannels? Clogging primarily occurs through two physical mechanisms:
- Arch Formation: Stable, static arches of particles block a channel constriction. This is common with solid particles and is largely independent of flow velocity and the constriction angle [1].
- Single-Particle Effects: Clogging caused by wall-particle adhesion or particle aggregation, which is more common in colloidal systems and with biological cells [1].
How does pulsatile flow help mitigate clogging, and what are the optimal parameters? Pulsatile flow can significantly delay clogging by introducing shear forces that prevent particle deposition and break apart aggregates. One study found that adding pulsations can achieve nearly a 100% improvement in filter half-life compared to steady flow [2].
- Key Parameters: Optimal mitigation requires specific combinations of amplitude and frequency.
- Optimal Setting: In one parallel microchannel system, an amplitude of 50% of the average driving pressure (150 mbar) at a frequency of 0.1 Hz nearly doubled the filter half-life [2].
- Critical Consideration: The frequency of pulsation must be compared to the average clogging rate of the specific system. Flow reversal can sometimes resuspend particles and accelerate the clogging of adjacent channels, so it is often advised to avoid it in parallel arrays [2].
Are there non-invasive, biocompatible methods to prevent clogging? Yes, emerging methods like 3D microbubble streaming offer a biocompatible solution. This technique involves trapping a microbubble in a cavity adjacent to a channel constriction and activating it with a piezotransducer. The resulting acoustic excitation generates counter-rotating vortices (microstreaming) that produce high shear stresses, inhibiting arch formation and disintegrating particle clusters in real-time [1]. This method is particularly promising for applications involving biological cells.
What is a simple, rapid method to clear a severely clogged chip? A simple, inexpensive protocol can revitalize chips clogged by cell clusters or polymer precipitation using a microwave oven [3].
- Flush the chip with a solvent (e.g., distilled water, ethanol, or acetone) using a syringe, applying as much hand pressure as possible.
- Remove all metal fittings (like hypodermic needles) from the chip.
- Place the chip in a standard kitchen microwave oven for 5 minutes at 500-700 watts.
- Remove the chip, reattach the ports, and flush with solvent again. Repeat the procedure if the first treatment doesn't fully clear the channels [3].
# Quantitative Data on Clogging Mitigation
Table 1: Comparison of Active Clogging Mitigation Techniques
| Technique | Mechanism of Action | Key Parameters | Reported Efficacy | Considerations |
|---|---|---|---|---|
| Pulsatile Flow [2] | High shear conditions erode particles and aggregates, rearranging filter cakes. | Amplitude, Frequency (e.g., 0.1 Hz) | Nearly 100% improvement in filter half-life | Can accelerate clogging if parameters cause flow reversal. |
| 3D Microbubble Streaming [1] | Acoustic actuation generates micro-vortices and shear stress to break clusters. | Actuation frequency, Bubble size, Cavity geometry | Real-time prevention and disintegration of clogs. | Biocompatible; can operate in event-triggered, continuous, or periodic modes. |
| Flow Resistance Design [4] | Introduces a controlled pressure drop to stabilize flow and reduce oscillations. | Channel width, depth, and length. | Prevents chip failure and connector leakage by predicting safe operating conditions. | A low-cost, passive method reliant on precise channel geometry. |
Table 2: Common Clogging Scenarios and Materials
| Clogging Scenario | Typical Culprits | Recommended Clearance Method | References |
|---|---|---|---|
| Cell Clustering | Aggregates of biological cells (e.g., in suspensions). | Microbubble streaming; Microwave protocol with water or ethanol. | [1] [3] |
| Polymer Precipitation | Hydrophobic polymers or solidified materials within channels. | Microwave protocol with solvents like acetone or isopropanol. | [3] |
| Solid Particle Aggregation | Polystyrene beads or other solid microparticles. | Pulsatile flow; Microbubble streaming. | [1] [2] |
# Experimental Protocols for Clogging Studies
Protocol 1: Establishing Pulsatile Flow to Delay Clogging
This protocol is adapted from studies using parallel microchannel arrays to investigate clogging dynamics [2].
Materials:
- Pressure Controller (e.g., Elveflow OB1 MK3+)
- Coriolis Flow Sensor
- Microfluidic chip with parallel channels
- Particle suspension (e.g., fluorescent polystyrene beads)
Methodology:
- Setup Configuration: Pressurize both the inlet and outlet reservoirs to limit bubble formation. Connect the pressure controller to the inlet and the flow sensor in series at the outlet to monitor flow rate.
- Baseline Measurement: Under steady flow conditions, record the baseline flow rate and the time until complete clogging.
- Pulsatile Flow Application: Use the pressure controller to generate a sinusoidal pressure profile at the inlet.
- Parameter Optimization: Systematically test different combinations of amplitude and frequency. A starting point is 50% of the average driving pressure at 0.1 Hz.
- Data Collection: Simultaneously record the flow rate and video footage of the channels. Correlate reductions in flow rate with specific clogging events observed visually.
- Efficacy Analysis: Compare the total processed volume and time until clogging against the baseline steady-flow experiment.
Protocol 2: Implementing Microbubble Streaming for Clog Prevention
This protocol outlines the use of integrated microbubbles for active clog mitigation [1].
Materials:
- PDMS microfluidic device with a dedicated lateral cavity for microbubble formation.
- Piezotransducer affixed to the microchip.
- Particle suspension.
Methodology:
- Device Priming: Rapidly infuse liquid into the microchannel to trap a gas pocket (e.g., air) in the lateral cavity, forming a quasi-cylindrical microbubble.
- Activation: Stimulate the piezotransducer near the bubble's resonant frequency to induce oscillatory fluid motion and generate steady 3D counter-rotating vortices (microstreaming).
- Operation Modes: The system can be run in:
- Continuous Mode: For constant prevention.
- Periodic Mode: To conserve energy.
- Event-Triggered Mode: Where a detected increase in pressure or reduction in flow triggers the activation.
- Validation: Use optical microscopy to confirm the disruption of particle arches and clusters near the constriction in real-time.
# Diagrams of Clogging Mechanisms and Solutions
Clogging Causes and Solutions
# The Scientist's Toolkit: Key Reagents & Materials
Table 3: Essential Research Reagents and Materials for Clogging Experiments
| Item | Specification / Example | Primary Function in Experiment |
|---|---|---|
| Fluorescent Particles | Polystyrene beads (e.g., 50 µm, 100 µm) [1] | Act as model cells/particles for visualizing clogging dynamics and testing mitigation. |
| Glycerol Solution | 23.8 w-w% in water [1] | Creates a neutrally buoyant medium for particles, preventing sedimentation-driven clogging. |
| PDMS Microfluidic Chips | Channels with constrictions (e.g., 50 µm to 10 µm) [2] | Provide the physical platform for creating and studying clogs at the microscale. |
| Piezotransducer | N/A | Affixed to the microchip to acoustically activate microbubbles and generate streaming flows [1]. |
| Pressure Controller & Flow Sensor | e.g., OB1 MK3+ & Coriolis sensor [2] | Enable precise generation of pulsatile flow and real-time monitoring of clogging via flow rate reduction. |
| Solvents for Clearing | Ethanol, Isopropanol, Acetone [3] | Used to flush and dissolve hydrophobic clogs (e.g., polymers) during clearance protocols. |
Cell-Adhesion Interactions and Surface Chemistry Effects
Troubleshooting Guides and FAQs
Frequently Asked Questions
- Q1: Why do my microfluidic channels keep clogging during single-cell analysis? A1: Clogging in microchannels often occurs due to the formation of stable arches or clusters of cells or particles at constrictions. This is analogous to a stable arch of granules blocking the neck of an hourglass. In colloidal systems like cell suspensions, clogging can also be caused by single-particle effects from wall-particle adhesion or particle aggregation [1].
- Q2: How can I prevent cells from adhering to my PDMS channel walls and causing blockages? A2: Cell adhesion is highly influenced by surface energy and roughness. Creating PDMS surfaces with moderate surface energy (approximately 70 mJ m⁻²) and an intermediate roughness ratio (approximately 2) can reduce uncontrolled adhesion. Furthermore, incorporating negatively charged groups (like sulfate groups) on particles or channel walls can create repulsive forces that prevent agglomeration and adhesion to PDMS walls [1] [5].
- Q3: Are there active methods to clear a clogged channel without disassembling my device? A3: Yes, active methods like 3D microbubble streaming can dislodge clogs in real-time. This technique involves activating a microbubble near a constriction with a piezotransducer. The bubble oscillation generates strong, 3D microstreaming vortices and significant shear stress that can break down particle clusters and inhibit arch formation, effectively preventing or clearing blockages [1].
- Q4: My single-cell encapsulation efficiency in droplets is low. How can I improve it? A4: In droplet microfluidics, single-cell encapsulation is a random process following a non-uniform Poisson distribution. To increase the proportion of droplets containing a single cell, you often need to highly dilute the cell suspension. However, this reduces throughput. Implementing a post-encapsulation sorting step, based on property differences, can help select and collect only the droplets that contain a single cell, thereby improving effective efficiency [6].
Troubleshooting Common Experimental Issues
Problem: Low or Dim Fluorescent Signal in Detection
- Potential Causes and Solutions:
- Reagent Quality: Check the storage conditions of your fluorescent antibodies or dyes. Reagents sensitive to improper storage may degrade. Visually inspect solutions for cloudiness or precipitation [7].
- Antibody Concentration: The concentration of your primary or secondary antibody may be too low. Consider testing a range of concentrations in parallel, changing only one variable at a time [7].
- Fixation and Washing: Inadequate fixation time or excessive rinsing during washing steps can diminish signal. Optimize fixation duration and the number of wash cycles [7].
- Microscope Settings: Verify the light source and settings on your microscope. This is the easiest variable to check first before re-running an experiment [7].
- General Workflow:
- Repeat the experiment to rule out simple human error.
- Ensure you have the appropriate positive and negative controls to confirm the protocol itself is functioning.
- Systematically check equipment and reagents.
- Change one variable at a time, documenting every modification meticulously [7].
- Potential Causes and Solutions:
Problem: Uncontrolled Cell Adhesion and Growth on Surfaces
- Potential Causes and Solutions:
- Substrate Surface Energy: Cell attachment, growth, and proliferation are highly dependent on the surface energy of the polymer substrate. Extremely high or low surface energy can suppress cell-surface interaction.
- Surface Roughness: Nanoscale topography provides mechanical anchoring for cells. An intermediate roughness ratio is often optimal.
- Experimental Protocol for Tuning PDMS Properties:
- Fabricate rough PDMS surfaces by partially curing a spun-coated PDMS layer, then exposing it to a candle flame to deposit a uniform carbon soot layer.
- Wash with a high-speed water jet to remove loosely bonded soot, creating a superhydrophobic surface with controlled roughness (5-150 nm) [5].
- Modify surface energy by exposing the superhydrophobic surfaces to oxygen plasma (12-30 W for 30 s to 2 min). This converts non-polar CH₃ groups to high-energy polar groups (like COOH and OH), making the surface hydrophilic and increasing its surface energy [5].
- Characterize the surface using a goniometer to measure water contact angle and a profilometer to measure roughness. The surface energy can be calculated using the OWRK method [5].
- Potential Causes and Solutions:
Table 1: Effect of PDMS Surface Properties on Cell Adhesion
| Surface Energy (mJ m⁻²) | Roughness Ratio | Water Contact Angle | Cell Adhesion Efficiency | Key Observation |
|---|---|---|---|---|
| ~21 | ~1.05 | ~110° | Low | Low-energy, smooth surface; poor for adhesion [5] |
| ~70 | ~2.0 | ~8° | High | Optimal conditions for efficient cell adhesion, growth, and proliferation [5] |
| ~100 | ~3.0 | ~0° | Low | High-energy, very rough surface; suppresses interaction [5] |
Table 2: Comparison of Microfluidic Anti-Clogging Techniques
| Technique | Mechanism | Key Advantage | Key Disadvantage |
|---|---|---|---|
| 3D Microbubble Streaming [1] | Acoustic activation of microbubbles generates shear stress and 3D vortices to break clusters. | Real-time, non-invasive clog prevention and clearance; biocompatible. | Requires integration of actuator and bubble cavity. |
| Passive Flow Loops/Spiral Channels [1] | Channel design induces recirculating Dean flows to keep particles suspended. | No external power required. | Less effective against strong clusters compared to active methods. |
| Pulsatile Flow [1] | Optimizing frequency and amplitude of unsteady flow delays clogging. | More effective than steady flow or simple flow reversal. | Requires a flow control system capable of generating pulses. |
| Dielectrophoresis [1] | Uses electrical fields to direct particle motion. | Effective for precise particle control. | Risk of cell damage; electrode fouling; complex and costly. |
Essential Research Reagent Solutions
Table 3: Key Materials and Reagents for Microfluidic Single-Cell Analysis
| Item | Function/Brief Explanation | Example Application |
|---|---|---|
| Polydimethylsiloxane (PDMS) [5] | Elastomeric polymer used to fabricate transparent, gas-permeable microfluidic channels via soft lithography. | Standard material for rapid prototyping of microfluidic devices. |
| Polystyrene Microspheres [1] | Suspended particles with tunable size and surface charge (e.g., sulfate groups), used to model cell behavior and test device performance. | Studying clogging dynamics and validating anti-clogging techniques in a controlled manner [1]. |
| Glycerol Solution [1] | Adjusts the density of the suspension medium to achieve neutral buoyancy for particles or cells. | Prevents sedimentation during experiments, ensuring particles remain suspended [1]. |
| Dulbecco's Modified Eagle Medium (DMEM) [5] | A standard cell culture medium containing nutrients, vitamins, and amino acids. | Supports the growth and proliferation of cells in vitro. |
| Fetal Bovine Serum (FBS) [5] | A common supplement to cell culture media, providing a rich source of growth factors and proteins. | Promoves cell health and adhesion in culture experiments. |
| Oxygen Plasma [5] | A treatment that functionalizes PDMS surfaces, creating polar groups that increase surface energy and wettability. | Converting hydrophobic PDMS channels to hydrophilic for improved fluidic control and altered cell-surface interactions [5]. |
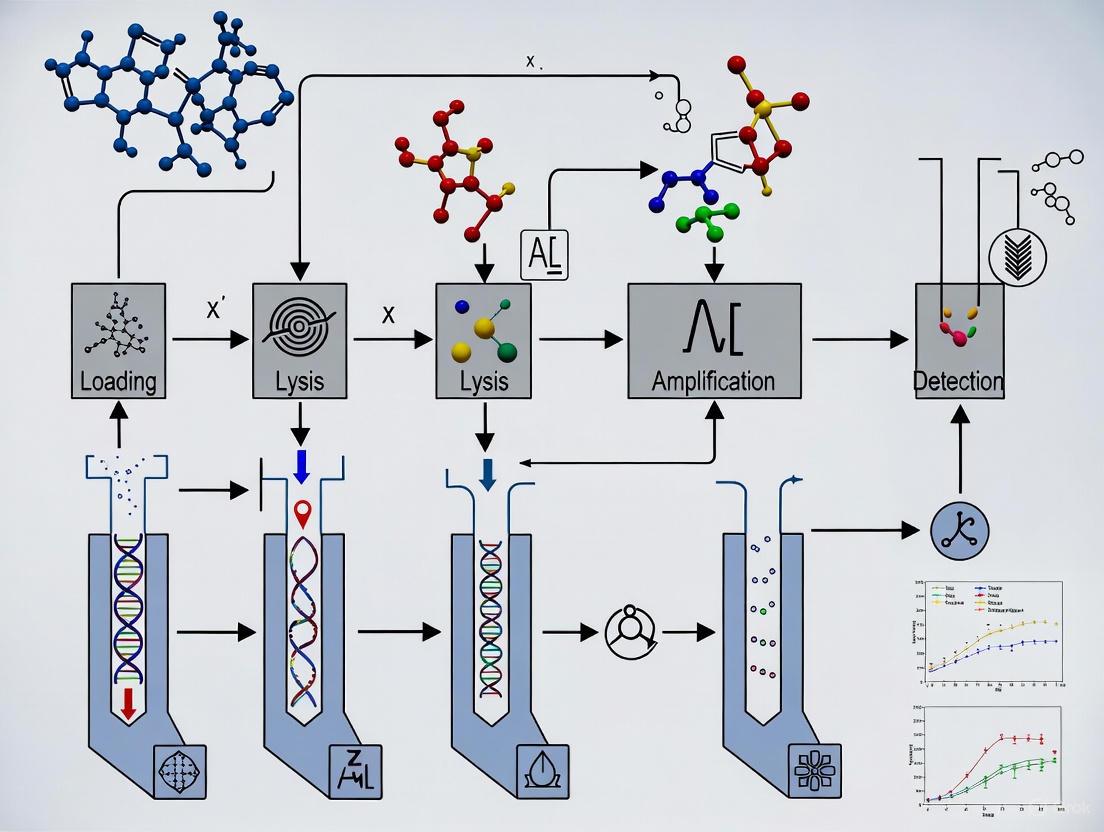
Experimental Workflow and System Diagrams
Microfluidic Clogging and Resolution Pathways
Single-Cell Analysis Workflow with Anti-Clogging
Impact of Particulate Contaminants and Sample Preparation Artifacts
Troubleshooting Guide & FAQs
Frequently Asked Questions
Q1: What are the most common causes of clogging in microfluidic devices for single-cell analysis? Clogging primarily occurs at channel constrictions where particles or cells form stable arches that block the flow. This is a universal challenge in systems where particles pass through narrow constrictions, observed in scenarios ranging from colloidal systems to microfluidic circuits. The main mechanisms involve single-particle effects due to wall-particle adhesion or particle aggregation, which are particularly problematic for biological cells in continuous systems [1].
Q2: How can I prevent clogging without complex hardware modifications? Applying controlled pulsatile flow is a highly effective strategy. Research shows that introducing a sinusoidal pressure profile at the inlet can significantly delay clogging. For instance, using a pressure amplitude of 50% of the average driving pressure (e.g., 150 mbar) at a frequency of 0.1 Hz can nearly double the filter half-life compared to steady flow conditions. It is generally advised to avoid flow reversal in parallel microchannels, as it may not improve performance [2].
Q3: Are there active methods to clear existing clogs? Yes, microbubble streaming is an innovative active method. By integrating a cavity near the channel constriction to trap a microbubble and activating it with a piezotransducer near its resonant frequency, you can generate 3D counter-rotating vortices (microstreaming). This creates high shear stress that disintegrates particle clusters and inhibits arch formation, effectively clearing blockages. The system can operate in event-triggered, continuous, or periodic modes to suit different application needs [1].
Q4: Can sample preparation itself introduce artifacts that affect analysis? Yes, sample preparation can introduce significant artifacts. In single-cell genomics, the time delay between sample extraction and processing can drastically alter transcriptome profiles. For example, storing Peripheral Blood Mononuclear Cells (PBMCs) at room temperature for over 2 hours initiates a time-dependent stress response, leading to a global downregulation of gene expression and a loss of immune cell identity markers. This sampling time effect can be a major driver of variance, surpassing even batch and donor effects in some cases [8].
Troubleshooting Common Issues
Problem: Rapid Clogging During Continuous Operation
- Symptoms: Sudden pressure increase, complete halt of flow, reduced experimental lifetime.
- Root Causes:
- Stable static particle arches at constrictions.
- Aggregation of biological cells or particles in the sample.
- High particle concentration leading to rapid accumulation.
- Solutions:
- Implement Pulsatile Flow: Use a pressure controller to generate a low-frequency oscillatory flow. This disrupts the formation of stable arches and can resuspend particles [2].
- Integrate Microbubble Streaming: Design your device with lateral cavities near constrictions to incorporate microbubbles. Acoustic actuation of these bubbles generates streaming vortices that prevent clogging [1].
- Apply Lateral Flow μ-Sieving: Combine a diagonal pillar filter with low-frequency mechanical oscillation (e.g., via a piezoelectric actuator). This mimics traditional sieving action, releasing smaller trapped particles from between larger ones and enabling continuous operation [9].
Problem: Low Yield or Poor Quality in Downstream Single-Cell Sequencing
- Symptoms: Low library yield, high duplicate rates, altered gene expression signatures in scRNA-seq data.
- Root Causes:
- Sampling time artifacts: Prolonged storage of cells at room temperature before processing [8].
- Cellular stress response impacting transcriptomes.
- Solutions:
- Minimize Processing Delay: Cryopreserve cells as quickly as possible after sample extraction. The ideal window is under 2 hours [8].
- Consider Cold Storage: For unavoidable delays, explore cold storage to minimize active cell stress responses [8].
- Computational Correction: For existing data, use a sampling time score based on a known signature of affected genes (e.g., upregulation of CIRBP and RBM3) to regress out this technical artifact [8].
- Experimental Correction: For some cell types, a short period of cell culture with specific activation (e.g., T-cell activation with anti-CD3) after thawing can reduce the sampling-induced artifact [8].
Quantitative Data on Clogging Mitigation Techniques
The table below summarizes the performance of different anti-clogging techniques as reported in the literature.
Table 1: Comparison of Anti-Clogging Techniques for Microfluidic Systems
| Technique | Key Operational Parameters | Reported Efficacy | Key Advantages |
|---|---|---|---|
| Pulsatile Flow [2] | Amplitude: 50% of average pressure (e.g., 75 mbar on 150 mbar). Frequency: 0.1 Hz. | Nearly 100% improvement in filter half-life. | Simplicity, can be implemented with standard pressure controllers. |
| Microbubble Streaming [1] | Activation near bubble resonant frequency. Operates in event-triggered, continuous, or periodic modes. | Effective real-time prevention and clearance of clogs. | Biocompatible, no moving parts, can be targeted to specific constrictions. |
| Lateral Flow μ-Sieving [9] | Low-frequency mechanical vibration applied to fluid. | 100% separation efficiency for target particles; >98% retrieval rate. | Prevents clogging and allows retrieval of filtered cells/particles. |
Experimental Protocols
Protocol 1: Implementing Pulsatile Flow for Clogging Mitigation
This protocol describes how to set up a pulsatile flow system to extend the operational life of a microfluidic device.
- Setup Configuration:
- Connect a pressure-driven flow controller (e.g., Elveflow OB1 MK3+) to the inlet of your microfluidic chip.
- Connect a flow sensor (e.g., Bronkhorst Coriolis) in series to monitor the flow rate.
- Pressurize both inlet and outlet reservoirs to improve gas solubility and limit bubble formation [2].
- Parameter Optimization:
- Set the average driving pressure according to your application (e.g., 150 mbar).
- Configure the controller to generate a sinusoidal pressure profile.
- Start with an amplitude of 50% of the average pressure and a frequency of 0.1 Hz.
- Correlate flow rate measurements with visual observations to fine-tune the amplitude and frequency for your specific system. Avoid parameters that cause flow reversal if using parallel channels [2].
- Operation:
- Initiate the pulsatile flow before introducing the particle/cell suspension and maintain it throughout the experiment.
Protocol 2: Minimizing Sampling Time Artifacts in Single-Cell Studies
This protocol outlines steps to prevent gene expression artifacts caused by delays in sample processing.
- Sample Collection:
- Collect blood or tissue samples under standardized conditions.
- Immediate Processing:
- Begin processing samples for single-cell analysis or cryopreservation immediately after extraction.
- The maximum recommended time at room temperature is 2 hours to prevent significant transcriptional changes [8].
- Cryopreservation:
- If immediate analysis is not possible, cryopreserve the isolated cells (e.g., PBMCs) without delay. Use controlled-rate freezing to maintain cell viability.
- Post-Thaw Processing (If Applicable):
- For certain applications, a short-term culture (e.g., 2 days) with cell-type-specific activation after thawing can help mitigate artifacts introduced during sampling delays [8].
Signaling Pathways and Workflows
Sampling Time-Induced Cellular Stress Pathway
The following diagram illustrates the cellular response to prolonged storage at room temperature, a key artifact in sample preparation.
Microbubble Streaming for Clog Prevention Workflow
This workflow shows how to integrate and use microbubble streaming to address clogging in a microfluidic device.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Key Materials and Reagents for Featured Experiments
| Item | Function / Application | Example from Literature |
|---|---|---|
| Polystyrene Microspheres | Used as model particles for testing and optimizing anti-clogging device performance. | Red fluorescent PS particles (50 and 100 µm) with sulfate groups for negative charge to prevent agglomeration [1]. |
| Glycerol Solution | Adjusts the density of the suspension medium to achieve neutral buoyancy for particles, promoting consistent flow behavior. | 23.8 w-w% glycerol in aqueous solution used to match particle density [1]. |
| PDMS (Polydimethylsiloxane) | The most common elastomer for rapid prototyping of microfluidic devices via soft lithography. | Used to create microchannels and device encapsulation [1] [9]. |
| SU-8 Photoresist | A negative, epoxy-based photoresist used to create high-aspect-ratio microstructures and molds for PDMS devices. | Spin-coated to a 30 µm thickness to define microfluidic channel patterns [9]. |
| Piezoelectric Actuator | Generates mechanical vibrations or acoustic excitation for active anti-clogging techniques. | Used to induce fluid oscillation in μ-sieving [9] and to excite microbubbles [1]. |
Consequences of Blockage on Throughput, Viability, and Analytical Reproducibility
Channel blockage is a significant technical challenge in microfluidic single-cell analysis, directly impacting the reliability and efficiency of research and diagnostic workflows. Clogging can occur from cell clusters, precipitated polymers, or other particulate matter within the microscale channels [10] [3]. This technical guide details the consequences of such blockages and provides established methodologies for prevention and resolution to ensure data integrity and operational continuity in your experiments.
Quantitative Impact of Blockage on Key Experimental Parameters
Blockages in microfluidic systems have direct and quantifiable negative effects on critical performance metrics. The table below summarizes the primary consequences.
Table 1: Consequences of Microfluidic Channel Blockage on Experimental Outcomes
| Performance Metric | Impact of Blockage | Underlying Cause | Effect on Data Quality |
|---|---|---|---|
| Throughput | Significant reduction or complete cessation of flow and cell processing [10]. | Physical obstruction of channels disrupts fluid dynamics and prevents cell passage. | Incomplete data acquisition; inability to process statistically relevant cell numbers [10]. |
| Cell Viability | Decreased viability due to increased shear stress and mechanical damage [11]. | Abnormal pressure buildup and shear forces around the obstruction damage cells. | Introduces bias; analysis may not reflect the native state of the cell population. |
| Analytical Reproducibility | Increased variability and loss of precision in single-cell analysis [10]. | Fluctuating flow rates and uneven distribution of cells and reagents. | Compromised reliability of results, such as single-cell RNA sequencing data [10] [12]. |
| Contamination Risk | Potential for cross-contamination between samples [10]. | Creation of "dead volume" where fluids can stagnate and mix. | False positive or false negative results in sensitive assays. |
Troubleshooting Guide: Blockage Prevention and Clearance
FAQ: Preventing Channel Blockage
Q1: What are the primary causes of clogging in microfluidic single-cell experiments? A: The main causes are:
- Cell Clusters: Incompletely dissociated tissues or cell cultures can form clusters larger than channel dimensions [3].
- Precipitated Polymers/Hydrogels: Polymers used in assays can precipitate out of solution within channels, especially if flow is interrupted [3].
- Complex Chip Architecture: Designs that integrate complex 3D fluidic networks, external pumps, and valves are particularly prone to creating dead volumes and clogging [10].
- Particulate Contaminants: Dust or impurities in samples or buffers.
Q2: How can I prevent blockages during experiment design? A: Implement these strategies:
- Robust Sample Preparation: Use rigorous filtration and single-cell suspension protocols to remove clusters and debris before loading [3].
- Optimized Channel Design: Where possible, utilize devices with simpler architectures or "active microfluidics" that use external fields (e.g., acoustic, electrical) for manipulation, reducing reliance on complex fluidic networks prone to clogging [10].
- Surface Treatment: Coat channel surfaces with additives like bovine serum albumin (BSA) or Pluronic to reduce cell and protein adhesion.
Experimental Protocol for Clearing a Clogged Chip
The following method provides a practical protocol for revitalizing a clogged microfluidic chip, adapted from a published technique [3].
Table 2: Research Reagent Solutions for Clog Clearance
| Item | Function/Explanation |
|---|---|
| 21 Gauge Hypodermic Needle & FEP Tube | To build a fluid-proof inlet port that can withstand high manual pressure. |
| 50 mL Syringe | For manual application of high pressure for flushing. |
| Filtered Distilled Water | Primary flushing solvent for general clogs. |
| Ethanol or Isopropanol | Flushing solvent for clogs caused by hydrophobic polymers or lipids [3]. |
| Microwave Oven | Applies heat to expand channel materials and dislodge obstructions. |
Workflow:
- Interface Setup: Build a tight, fluid-proof inlet by fitting a FEP (fluorinated ethylene-propylene) tube over a 21-gauge hypodermic needle, which is attached to a 50 mL syringe.
- Flushing: Identify the clog location via microscopy. Insert the FEP tube into the port farthest from the clog. Flush the chip manually with filtered distilled water, applying as much pressure as possible. For hydrophobic clogs (e.g., polymers), use ethanol, isopropanol, or acetone.
- Microwave Treatment: Remove all metal parts (e.g., the needle) from the chip. Place the chip in a standard kitchen microwave oven and heat for 5 minutes at 500-700 watts. The thermal expansion can help dislodge the clog.
- Post-Treatment Flushing: Reinstall the port and immediately flush the chip again with the appropriate solvent.
- Repetition: If one treatment cycle does not clear the clog, repeat the entire procedure [3].
This workflow for clearing a clogged chip can be visualized as a two-stage process, as shown in the following diagram.
Integrated Strategies for Blockage-Free Single-Cell Analysis
Achieving robust and reproducible results requires an integrated approach that combines device selection, experimental design, and operational protocols. The following diagram illustrates the logical relationship between overarching goals and specific technical strategies to prevent blockages.
Key Integrated Considerations:
- Technology Selection: When designing new experiments, consider emerging active microfluidic platforms. These systems use external fields (e.g., electrical, magnetic, acoustic, optical) to manipulate single cells, often eliminating the need for complex microchannel networks that are prone to clogging [10].
- Systematic Sample Handling: The foundation of blockage prevention lies in sample quality. Establish and validate standard operating procedures (SOPs) for cell sample preparation, including filtration steps and visual inspection for aggregates immediately before loading.
- Proactive Maintenance: Incorporate the flushing and clearance protocols outlined in this guide as routine maintenance steps, not just emergency responses, to ensure consistent device performance over time.
Evaluating Economic and Operational Costs of System Downtime
FAQs: Understanding and Quantifying Downtime
What is the immediate financial impact of system downtime? The immediate cost is often calculated as lost revenue. A widely cited industry average puts the cost of IT downtime at approximately $5,600 per minute, though this can range from $137 to $427 per minute for small businesses and up to $9,000 per minute for larger enterprises [13]. You can estimate this using the formula: Downtime Cost = (Revenue per hour / Operating hours) x Downtime hours [14].
Beyond lost revenue, what other costs should a research lab consider? Lost revenue is just one component. The full cost includes [13] [15] [14]:
- Business Disruption: This is often the largest cost share, including reputational damage, loss of future grant funding, and delays in publication.
- Lost Productivity: Salaries for researchers and technicians who cannot perform their work, plus overtime costs to catch up.
- Regulatory and Compliance Fines: Particularly relevant in clinical or GxP-compliant research environments.
- Equipment and Repair Costs: Emergency servicing, replacement of damaged components (e.g., a burst microfluidic chip), and depleted reagents.
How does the business model affect potential losses? Organizations whose core operations rely on continuous system access face the highest risks. In a research context, a core facility that provides fee-for-service single-cell RNA sequencing has a business model similar to e-commerce, where an outage directly halts revenue generation [13].
What is a Business Impact Analysis (BIA) and how can it help our lab? A Business Impact Analysis (BIA) is a formal process that predicts the consequences of disrupting business operations and gathers information needed to develop recovery strategies [15]. For a research lab, conducting a BIA involves [15] [16]:
- Identifying critical processes (e.g., single-cell encapsulation, library preparation).
- Determining the operational and financial impacts of their interruption.
- Establishing the Maximum Tolerable Downtime (MTD) for each process.
- Using this data to prioritize recovery efforts and justify investments in prevention.
Troubleshooting Guide: Pressure Instability and Channel Blockage in Microfluidics
A primary cause of downtime in microfluidic single-cell analysis is abnormal pressure and flow, often leading to channel blockage. The following guide helps diagnose and resolve these issues.
Pressure Troubleshooting Flowchart
The diagram below outlines a logical workflow for diagnosing common pressure-related problems in microfluidic systems [4] [17].
Establishing a Pressure Baseline
Before troubleshooting, establish a "system reference pressure" for your setup. Use a standard chip geometry and an easy-to-replicate fluid (e.g., PBS) at a set flow rate and temperature. Record the pressure under these conditions for future comparison [17].
Understanding Pressure-Flow Relationships
In microfluidics, the pressure drop (Δp) is related to the flow rate (Q) by the hydrodynamic resistance (RH). For a rectangular channel, this relationship is defined by [4]:
Δp = RH · Q
Where the hydrodynamic resistance is calculated as:
RH = (12μL) / (W · H · [min(W,H)]² · [1 - 0.6274ε · tanh(π/(2ε))])
- μ: Dynamic viscosity of the fluid
- L: Channel length
- W: Channel width
- H: Channel depth
- ε: min(W/H, H/W)
This confirms that for a given geometry and fluid, flow rate (Q) is linearly proportional to the applied pressure drop (Δp) under laminar flow conditions [4]. Understanding this helps predict how changes in channel design or fluid properties will affect operating pressure.
The Scientist's Toolkit: Key Research Reagent Solutions
The table below lists essential materials for microfluidic experiments aimed at preventing downtime due to blockage.
| Item | Function | Application Note |
|---|---|---|
| In-line Frit/Filter | A 0.5-µm or 0.2-µm porosity frit placed after the autosampler or sample loader. It acts as a sacrificial element, trapping debris from samples before it reaches the microfluidic chip. It is inexpensive and easy to replace, protecting the expensive chip from damage [17]. | Use a 0.5-µm frit for channels >2 µm; a 0.2-µm frit for channels ≤2 µm [17]. |
| Pressurized Syringe System | A low-cost alternative to commercial pumps. Provides a stable pressure source for flow control when calibrated against channel geometry-specific hydrodynamic resistance [4]. | Ideal for resource-limited labs. Requires prior calibration of the Δp-Q relationship for your specific chip design to prevent over-pressure failure [4]. |
| Hollow Metal Connectors & Silicone Tubing | Inexpensive, robust connection hardware for linking syringes or pressure sources to the microfluidic chip. Polyetheretherketone (PEEK) accessories are often costly and unnecessary for most aqueous applications [4]. | Silicone, PMMA, PDMS, PET, or polycarbonate are often sufficient and more affordable materials for tubing and connectors [4]. |
| Hydrodynamic Resistance Calculator | A script or software tool that implements the RH formula. It allows researchers to pre-determine safe operating pressures (Δp) for a desired flow rate (Q) based on their channel's width, depth, and length, preventing chip failure [4]. | A critical design tool for predicting achievable flow regimes and ensuring operational stability. |
Advanced Microfluidic Platforms and Anti-Clogging Design Methodologies
Troubleshooting FAQs
Q1: How can I prevent clogging in the narrow channels of my droplet generation device?
A: Clogging, often caused by cell clusters or stable static arches at constrictions, can be mitigated through several active and passive strategies [1]:
- Microbubble Streaming: Integrate a lateral cavity near the channel constriction to trap a microbubble. When activated by a piezotransducer, this creates counter-rotating vortices (microstreaming) that disrupt arch formation and break apart cell clusters. This method can operate in event-triggered, continuous, or periodic modes [1].
- Optimized Channel Geometry: Use channel constrictions with a neck-to-particle size ratio (W/d) of at least 1.5 to reduce the probability of clogging [1].
- Pulsatile Flow: Applying oscillatory or pulsatile flow has been shown to delay microchannel clogging compared to steady flow conditions [1].
- On-Chip Sample Enrichment: Implement a device that focuses cells and then removes excess aqueous phase before droplet generation. This allows you to use a lower initial cell concentration, which reduces cell-cell interactions and the risk of clogging in the narrow focusing unit [18].
Q2: My single-cell encapsulation rate is low. What strategies can improve it?
A: Low encapsulation rates often stem from the inherent randomness of Poisson distribution. Moving to deterministic, active, or passive hydrodynamic focusing can significantly enhance efficiency [19] [18] [20].
- Passive Hydrodynamic Trapping: Utilize devices based on the "least flow resistance path" principle. These designs use concatenated T and inverse T junction pairs to guide cells sequentially into trap sites, achieving up to 90-100% single-cell trapping efficiency [20].
- Inertial Focusing with Sample Enrichment: A device with a double spiral channel can inertially focus cells into a single file. An integrated sample enrichment module then removes excess medium, increasing the effective cell concentration just before encapsulation. This method has achieved single-cell encapsulation rates of 72.2% for cells and 79.2% for beads [18].
- Avoid High-Censity Suspensions: High cell concentrations can hinder focusing efficiency in spiral channels due to cell-cell interactions. Starting with a lower density (e.g., (2 \times 10^6) cells/mL) and enriching on-chip is more effective [18].
Q3: How do I select the right surfactant for my water-in-oil (W/O) droplet application?
A: Surfactant selection is critical for stable droplet generation, preventing coalescence, and ensuring biocompatibility [21].
- Emulsion Type: For W/O emulsions, you need a lipophilic surfactant with a low Hydrophilic-Lipophilic Balance (HLB) value, typically below 7.
- Key Properties: Consider the Critical Micelle Concentration (CMC) and interfacial tension. A lower interfacial tension (e.g., 4 mN/m) generally facilitates easier droplet generation.
- Application-Specific Formulations: For sensitive biological applications like single-cell analysis or ddPCR, fluorinated surfactants like the FluoSurf family are ideal due to their high stability, biocompatibility, and purity. FluoSurf-O is specifically designed for fluorescence-based applications requiring ultra-low autofluorescence [21].
The table below compares common and specialized surfactants for W/O droplet generation.
Table 1: Surfactant Selection Guide for Water-in-Oil (W/O) Droplet Generation
| Surfactant | Type | HLB Value | Key Features | Ideal For |
|---|---|---|---|---|
| Span 80 | Nonionic | ~4.3 | Common, cost-effective; provides steric stabilization [21]. | General W/O emulsification, food, and pharmaceuticals [21]. |
| PGPR | Nonionic | ~3.5 | Highly effective in stabilizing high internal phase emulsions [21]. | Food (chocolate), drug delivery systems [21]. |
| FluoSurf-C | Fluorinated Nonionic | 1.5 | High stability in fluorinated oils; reliable for droplet formation [21]. | General ddPCR, single-cell analysis [21]. |
| FluoSurf-O | Fluorinated Nonionic | 1.5 | Ultra-low autofluorescence [21]. | Fluorescence detection, sensitive optical measurements [21]. |
| FluoSurf-S | Fluorinated Nonionic | 3.9 | Lower molecular weight; stable under thermocycling [21]. | High-frequency droplet gen., ddPCR with viscous solutions [21]. |
Q4: What are the common causes of droplet instability and how can I improve it?
A: Droplet instability, including coalescence (merging) or Ostwald ripening (shrinkage/growth), is primarily a surfactant-related issue [21].
- Incorrect Surfactant Type/Concentration: Using a surfactant with the wrong HLB value or a concentration below its Critical Micelle Concentration (CMC) will not sufficiently lower interfacial tension, leading to coalescence. Always use a surfactant concentration above its CMC.
- Insufficient Stabilization: Ensure the surfactant can form a stable film around the droplet. For fluorinated oil systems, fluorinated surfactants like FluoSurf are most effective.
- Biocompatibility Conflicts: Some biological components (e.g., proteins, cells) can interfere with surfactant function. Choose biocompatible surfactants like Pluronic F-68 (for O/W) or FluoSurf (for W/O) for cell-based applications [21].
Detailed Experimental Protocols
Protocol 1: High-Efficiency Single-Cell Encapsulation via On-Chip Sample Enrichment
This protocol is adapted from a study that achieved a 72.2% single-cell encapsulation rate using a double spiral microfluidic device with integrated sample enrichment [18].
1. Device Fabrication:
- Design: The PDMS chip comprises three units: (1) an 8-loop double spiral focusing unit (channel width: 100 μm, depth: 60 μm) with half-circle pillars on the inner curve, (2) a flow resistance-based sample enrichment module with five serpentine units, and (3) a standard cross-flow droplet generation unit [18].
- Molding: Fabricate an SU-8 master mold using standard photolithography.
- PDMS Casting & Bonding: Cast PDMS (Sylgard 184) at a 10:1 base-to-curing agent ratio onto the mold. After cross-linking, punch inlets/outlets and bond to a flat PDMS slab using oxygen plasma [18].
- Surface Treatment: Treat the channels with a fluorosilane (e.g., 1H,1H,2H,2H-perfluorododecyltrichlorosilane) to render them hydrophobic and prevent aqueous phase wetting. Bake and optionally treat with a commercial superhydrophobic solution before use [18].
2. Sample Preparation:
- Culture and harvest your cells (e.g., MDA-MB-231). Digest, centrifuge, and resuspend them in culture medium at a density of (2 \times 10^6) cells/mL [18].
- Prepare the continuous phase, e.g., fluorocarbon oil (Novec 7500) containing 2% (w/w) surfactant (e.g., Pico-Surf) [18].
3. Experimental Setup & Operation:
- Connect syringes containing the cell suspension and oil to the chip inlets via tubing.
- Mount the chip on an inverted microscope equipped with a high-speed camera.
- Set the aqueous phase (cell suspension) flow rate to 80 μL/min and the oil phase flow rate to 70 μL/min using syringe pumps [18].
- Initiate the flows. Cells will be focused into a single file in the double spiral. The enrichment module will then remove over 50% of the aqueous phase, increasing the effective cell density before droplet generation at the cross-junction [18].
4. Data Analysis:
- Record the droplet generation process at the junction (e.g., at 2000 fps).
- Use a droplet detection algorithm (e.g., based on YOLOv8n) to analyze the videos and count the number of cells per droplet to statistically determine the encapsulation rate [18].
Table 2: Key Parameters for High-Efficiency Encapsulation Protocol [18]
| Parameter | Specification | Effect |
|---|---|---|
| Initial Cell Density | (2 \times 10^6) cells/mL | Reduces cell-cell interactions, improves focusing. |
| Spiral Channel Width | 100 μm | Balances focusing efficiency and clogging risk. |
| Aqueous Flow Rate | 80 μL/min | Optimized for focusing and enrichment in this device. |
| Oil Flow Rate | 70 μL/min | Produces stable droplets at ~4000 droplets/second. |
| Single-Cell Encapsulation Rate | 72.2% (cells), 79.2% (beads) | Significantly higher than Poisson-limited (~37%) random encapsulation. |
Protocol 2: Anti-Clogging via Microbubble Streaming
This protocol describes the integration and use of 3D microbubble streaming to prevent clogs at channel constrictions [1].
1. Device Fabrication with Microbubble Cavity:
- Fabricate a straight PDMS microchannel (e.g., 20 mm long) with a rectangular cross-section (e.g., 450 μm x 150 μm) that narrows linearly (e.g., at a 45° angle) to a constriction (e.g., 150 μm x 150 μm) [1].
- Critical Step: Design a lateral cavity (e.g., 80 μm wide, 500 μm long) adjacent to the channel, just before or at the constriction. This cavity will trap the microbubble [1].
2. System Setup:
- Connect the microfluidic device to syringe pumps for fluid control.
- Affix a piezotransducer to the microchip, close to the bubble cavity, and connect it to a function generator and amplifier for acoustic actuation [1].
- Mount the entire setup on a vibration-damped table.
3. Operation:
- Prime the device with your particle or cell suspension. A gas (e.g., air) pocket will be trapped in the lateral cavity, forming a quasi-cylindrical microbubble [1].
- Triggering Modes: Activate the piezotransducer near the bubble's resonant frequency. You can choose from three operational modes:
- Event-Triggered: Activate only when a clog is detected.
- Continuous: Run streaming continuously for persistent challenges.
- Periodic: Activate at set intervals for preventative maintenance [1].
- The generated 3D microstreaming vortices will disrupt particle arches and clusters, preventing or clearing blockages in real-time [1].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Droplet Microfluidics Experiments
| Item | Function / Description | Example Products / Notes |
|---|---|---|
| Fluorinated Oil | Continuous phase for W/O emulsions; immiscible with aqueous cell suspension. | Novec 7500, Fluo-Oil 40, Fluo-Oil 7500 [18] [21]. |
| Fluorinated Surfactant | Stabilizes aqueous droplets in fluorinated oil; prevents coalescence and leakage. | FluoSurf range (Emulseo) [21]. |
| Cell-Permeable Surfactant | For lysis buffer in Drop-Seq; lyses cells inside droplets to release mRNA. | Custom lysis buffer with compatible surfactant [22]. |
| Barcoded Beads | Microparticles with primers for mRNA capture in single-cell sequencing; contain cell barcodes and UMIs. | Chemically synthesized beads or hydrogel beads for Drop-Seq [22]. |
| PDMS | Silicone elastomer used to fabricate flexible, transparent microfluidic chips. | Sylgard 184 [18]. |
| Surface Treatment | Chemicals that render channel walls hydrophobic, crucial for W/O droplet formation. | Fluorosilanes, MesoPhobic-2000 [18]. |
Workflow and Decision Diagrams
Diagram 1: Single-Cell Encapsulation Strategy
Diagram 2: Surfactant Selection Logic
Troubleshooting Guides & FAQs
Frequently Asked Questions
Q1: What are the most common causes of slow or erratic movement in a pneumatic actuator?
A: Slow or erratic actuator movement can stem from several sources:
- Excessive air choke: This can be due to an incorrectly adjusted flow control valve, a plugged air filter or silencer, or a squeezed tube [23].
- Pressure too low: This may be caused by a damaged regulator, a plugged filter, system leaks, or a stuck valve [23].
- Load misalignment: If the load is misaligned with the cylinder, it can cause erratic movement and increased friction. Re-aligning the cylinder and load is the recommended action [23].
- Internal leaks: Worn piston seals can cause drift and slow movement. To check, pressurize one side of the cylinder and observe for leakage from the opposite side [23].
Q2: My directional control valve is not changing position. What should I check?
A: Start with these troubleshooting steps:
- Check the electrical supply: An electric failure can prevent the solenoid coil from picking up. Fix the electrical issue and then check if the spool moves manually [23].
- Inspect the valve spool: The spool can become stuck due to impurities between the spool and its sleeve, or a failed pilot. This may require cleaning or replacing the valve [23].
- For proportional valves: Verify that the valve is receiving the correct set value from the control program. If it is receiving the signal but not responding, the valve itself may need replacement [23].
Q3: How can I intentionally reduce the speed of a pneumatic actuator for a controlled process?
A: The most common and effective method is to use flow control valves [24].
- Metering Out: Restricting the air exhausting from the cylinder is the preferred technique. It provides smoother control by creating back pressure on the exiting side of the piston [24].
- Metering In: Restricting the air entering the cylinder is also possible but can lead to less stable, "spongy" motion under varying loads [24].
- Other methods include reducing the system's supply pressure (though this also reduces force) or using smaller diameter tubing to restrict flow [24].
Q4: I have a continuous air leak from the vent hole on my regulator. What does this indicate?
A: A continuous leak from the regulator's vent hole typically indicates a faulty main bonnet or a damaged diaphragm [23]. You should schedule a repair immediately. A complete diaphragm failure could apply full system pressure downstream, potentially causing solenoid valves to shift unexpectedly and creating a safety hazard [23].
Q5: What is an active method to prevent clogging in microfluidic channels used for single-cell analysis?
A: An innovative strategy is to use 3D microbubble streaming [1]. This method involves trapping a microbubble in a cavity adjacent to a channel constriction and activating it with a piezotransducer. The bubble oscillation generates strong, 3D microstreaming vortices that produce high shear stress, effectively disrupting particle arches and clusters before they can form a stable clog [1]. This is a biocompatible and effective anti-clogging technique for continuous microfluidic operations.
Troubleshooting Guide: Common Pneumatic System Issues
| Problem Category | Specific Symptom | Possible Cause | Recommended Action |
|---|---|---|---|
| Actuator Performance | Moving abnormally slow | Plugged filter [23], Incorrectly adjusted flow control valve [23], System air leaks [23] [25], Low supply pressure [23] | Replace filter [23], Readjust flow control valve [23], Fix leaks using soapy water to identify sources [25], Check and adjust regulator [23] |
| Erratic or jerky movement | Load misalignment [23], Lack of lubrication [25], Stuck or malfunctioning valve [25] | Re-align cylinder and load [23], Ensure proper system lubrication [25], Clean or replace the valve [25] | |
| Pressure Issues | Pressure drops in system | Dirty or clogged filters [23] [25], Air leaks [25], Undersized compressor [25] | Clean or replace filters [23] [25], Identify and fix leaks [25], Ensure compressor provides adequate pressure and volume [25] |
| Air leak at regulator vent | Damaged diaphragm or seals [23] | Replace diaphragm and seals using an overhaul kit as soon as possible [23] | |
| Valve Operation | Directional valve not shifting | Electric failure (solenoid coil) [23], Valve spool stuck due to dirt [23] | Fix electrical connection and check voltage [23], Clean or replace the valve [23] |
| Solenoid coil burnout | Low voltage [23], Voltage transients [23], Valve spool stuck preventing armature seating [23] | Check for high-resistance connections and transformer voltage [23], Isolate solenoid circuits and use electrical filters [23], Repair or replace stuck valve [23] | |
| Air Preparation | Contaminated air downstream | Over-contaminated filter element [23], Condensate in filter bowl [23] | Replace filter element regularly [23], Drain condensate frequently; install an automatic drain if needed [23] |
| Cylinder Issues | Cylinder drift from position | Piston seal leak [23], Internal leak in directional control valve [23] | Replace piston seals [23], Repair or replace the directional control valve [23] |
| Cylinder body or rod seal leak | Loose tie rod, pinched seal, or seal deterioration [23] | Tighten tie rods or replace damaged seals [23] |
Experimental Protocol: Anti-Clogging via Microbubble Streaming
This protocol details the methodology for employing 3D microbubble streaming to prevent clogging in microfluidic channels, as derived from relevant scientific literature [1].
1. Objective: To prevent real-time clogging and disrupt particle clusters at microchannel constrictions using acoustically-activated microbubbles.
2. Materials & Equipment:
- Microfluidic Chip: Fabricated via soft lithography, featuring a main channel with a constriction and an adjacent lateral cavity for microbubble formation [1].
- Piezoelectric Transducer (Piezotransducer): Affixed to the microchip to provide acoustic excitation [1].
- Function Generator & Amplifier: To generate and amplify the electrical signal for driving the piezotransducer.
- Optical Setup: Including an inverted microscope, high-speed camera, and vibration-damped table for flow visualization and data capture [1].
- Sample: A suspension of particles (e.g., 50 or 100 µm fluorescent polystyrene particles) in an aqueous glycerol solution adjusted for neutral buoyancy [1].
- Syringe Pump: For controlling the flow of the particle suspension through the microchannel.
3. Procedure: 1. Chip Priming: Fill the microfluidic channel with the carrier fluid, ensuring a single, quasi-cylindrical air bubble is trapped in the lateral cavity near the constriction [1]. 2. System Setup: Connect the piezotransducer to the function generator and amplifier. Mount the microfluidic chip on the microscope stage and connect it to the syringe pump loaded with the particle suspension. 3. Flow Initiation: Start the syringe pump to introduce the particle suspension into the main channel at a predetermined, constant flow rate. 4. Activation & Monitoring: * Event-Triggered Mode: Monitor the constriction via camera. Upon detection of a particle cluster or the initiation of an arch, immediately trigger the piezotransducer. * Continuous/Periodic Mode: Activate the piezotransducer continuously or at set intervals throughout the experiment. 5. Acoustic Actuation: Drive the piezotransducer at a predetermined frequency (typically near the resonant frequency of the microbubble) and amplitude to induce vigorous bubble oscillation [1]. 6. Data Collection: Record the flow dynamics at the constriction before, during, and after actuation to quantify the efficacy in disrupting clusters and preventing clogs.
4. Data Analysis:
- Compare the frequency and duration of clogging events between experiments with the microbubble activated versus deactivated.
- Use statistical analyses to validate the method's effectiveness under different particle sizes, flow conditions, and operational modes [1].
Quantitative Data for Microfluidic Anti-Clogging Experiments
The table below summarizes key parameters from experimental research on microbubble streaming for anti-clogging, providing a reference for your own experimental design [1].
| Parameter Category | Specific Parameter | Value / Description |
|---|---|---|
| Microchannel Geometry | Main Channel Cross-Section | 150 µm (H) x 450 µm (W) [1] |
| Constriction Cross-Section | 150 µm (H) x 150 µm (W) [1] | |
| Convergence Angle | 45° [1] | |
| Microbubble Cavity Dimensions | 500 µm (length) x 80 µm (width) [1] | |
| Particle Specifications | Material | Polystyrene, fluorescent [1] |
| Sizes Used | 50 µm & 100 µm [1] | |
| Neck-to-Particle Size Ratio (W/d) | 3 (for 50µm) & 1.5 (for 100µm) [1] | |
| Surface Charge | Negatively charged sulfate groups [1] | |
| Carrier Fluid | Aqueous solution with 23.8 w-w% glycerol [1] | |
| Operational Modes | Activation Modes | Event-triggered, Continuous, or Periodic [1] |
Research Reagent Solutions
This table lists essential materials and their functions for setting up a microfluidic system with pneumatic actuation and anti-clogging features.
| Item | Function / Application |
|---|---|
| Polydimethylsiloxane (PDMS) | A transparent, durable elastomer used for rapid prototyping of microfluidic chips via soft lithography [1]. |
| Fluorescent Polystyrene Particles | Used as cell analogs or model particles for visualizing flow, testing clogging behavior, and calibrating system performance [1]. |
| Aqueous Glycerol Solution | Serves as a carrier fluid; the glycerol concentration can be adjusted to match particle density, achieving neutral buoyancy to mitigate sedimentation [1]. |
| Piezoelectric Transducer | Converts electrical signals into mechanical vibrations to acoustically excite microbubbles, generating the required microstreaming flows [1]. |
| Compressed Air or Inert Gas (e.g., Argon) | The power source for pneumatic valves and actuators. Inert gases can be used for microbubble formation when interaction with atmospheric air must be avoided [1]. |
| Solenoid Valve | An electrically-operated valve that provides fast and precise control over pneumatic pressure to actuate control lines on a microfluidic chip [23]. |
| Filter-Regulator-Lubricator (FRL) Unit | Conditions compressed air: the filter removes contaminants, the regulator sets working pressure, and the lubricator introduces lubricant to reduce friction in pneumatic components [23]. |
System Integration and Workflow Diagrams
Anti-Clogging Control Logic
Pneumatic Control System Integration
This technical support center article provides troubleshooting and methodological guidance for researchers working with hydrodynamic traps in microfluidic devices. A primary challenge in this field is channel blockage, which can compromise high-throughput single-cell analysis. This content, framed within a broader thesis on preventing such blockages, offers solutions grounded in specific trap geometries and operational protocols. The following sections detail common issues and provide verified experimental methods to optimize your system's performance.
Troubleshooting Guides
Problem 1: Low Single-Cell Trapping Efficiency
Issue: The device traps multiple cells per site or leaves many trap sites empty.
| Possible Cause | Diagnostic Steps | Recommended Solution |
|---|---|---|
| Suboptimal Flow Resistance Ratio | Use CFD simulation to calculate the flow rate ratio (Q1/Q2) between the trapping path and the bypass channel. [20] | Optimize the geometry of concatenated T-junction pairs to ensure the Q-ratio is greater than 1 when a trap is empty, and less than 1 when occupied. [20] |
| Incorrect Trap Size | Measure the diameter of your target cells and the narrowest constriction of the trap. | Design trap constrictions and pockets relative to cell size. For example, one device for 4µm yeast cells used 10µm x 10µm V-shaped pockets with 2µm openings. [26] |
| High Cell Concentration | Check the cell density of the loaded suspension under a microscope. | Dilute the cell suspension to prevent overcrowding and ensure a sequential, deterministic trapping process. [20] |
Problem 2: Clogging and Cluster Formation in Microchannels
Issue: Blockages form at constrictions, halting flow and trapping operations.
| Possible Cause | Diagnostic Steps | Recommended Solution |
|---|---|---|
| Stable Arch Formation | Visually inspect if particles form a stable arch at the channel entrance. | Integrate microbubble streaming. Activate a piezotransducer near the constriction to generate 3D counter-rotating vortices that disrupt arch formation. [1] |
| Particle Adhesion/Aggregation | Check for cluster formation in the sample reservoir or channel upstream. | Use negatively charged particles/cells or include stabilizers in the suspension to reduce agglomeration. [1] Implement pulsatile flow instead of steady flow to delay clogging. [1] |
| Insufficient Clog Removal | Monitor if clogs persist despite continuous flow. | Employ an event-triggered or periodic anti-clogging system. Use a control system to activate microbubble streaming only when a clog is detected or at set intervals. [1] |
Problem 3: Low Cell Viability Post-Trapping
Issue: Cells are damaged during the trapping process or show poor health during subsequent culture.
| Possible Cause | Diagnostic Steps | Recommended Solution |
|---|---|---|
| Excessive Shear Stress | Calculate fluid shear stress at trap sites using CFD, particularly in narrow constrictions. | Redesign trap geometry to incorporate smooth, arc-contoured transitions rather than sharp corners to reduce shear forces. [20] |
| Prolonged Trapping Time | Time the entire process from cell loading to the start of the assay. | Use a compact trap configuration (e.g., T-junction pairs) to reduce the flowing path of cells, decreasing the total trapping operation time. [20] |
| Long-Term Culture Challenges | Check for adequate nutrient delivery and waste removal in the trap. | For long-term studies, ensure the trap design allows for continuous, low-flow-rate perfusion of fresh medium to maintain the cells. [26] |
Frequently Asked Questions (FAQs)
Q1: What is the core hydrodynamic principle used in these high-efficiency traps? The fundamental principle is the "least flow resistance path." An empty trap is designed to have lower flow resistance than its adjacent bypass channel, directing the cell-carrying fluid into the trap. Once a cell is trapped, it significantly increases the flow resistance of that path. The fluid then diverts to the next empty trap with the lowest resistance, enabling sequential and deterministic loading. [20]
Q2: How can I save space on my chip while maintaining a high density of trap sites? Replace long, serpentine main channels with a series of concatenated T and inverse T junction pairs. This compact configuration was shown to save approximately 2-fold in space compared to earlier designs, allowing for thousands of trap sites on a 1-cm² area. [20]
Q3: Our lab is new to microfluidics. What is a simple trap geometry we can start with? A V-shaped pocket design is a robust starting point. It uses hydrodynamic forces to guide and hold cells in pre-defined positions. One proven design connects a 20µm wide main channel to 10µm x 10µm V-shaped pockets via 2µm wide confinement openings, successfully used for trapping yeast cells. [26]
Q4: Are there active methods to prevent clogging without redesigning the entire chip? Yes, integrating 3D microbubble streaming is a highly effective active method. By placing a microbubble-containing cavity adjacent to a constriction and activating it with a piezotransducer, you can generate strong fluid vortices that prevent arch formation and break apart clusters in real-time. [1]
Q5: What quantitative metrics should I use to validate my device's performance? Key performance indicators include:
- Single-Cell Trapping Efficiency: The percentage of occupied traps that contain exactly one cell. Aim for 90% or higher. [20]
- Trapping Speed: The time required to fill all trap sites. Advanced designs can fill 400 sites in under 10 minutes. [20]
- Cell Viability Post-Trapping: The percentage of trapped cells that remain viable, which should be high (e.g., >97% directly after capture). [27]
Experimental Protocols & Data
Protocol 1: Implementing a Deterministic Hydrodynamic Trap
This protocol is based on a device that achieved 100% cell trapping and 90% single-cell trapping efficiency over 400 sites. [20]
1. Design and Fabrication:
- Core Structure: Design the trapping unit with concatenated T and inverse T junction pairs. The main channel should have a constant geometry, while the trap constrictions are narrower.
- CFD Simulation: Before fabrication, use Computational Fluid Dynamics (CFD) software to simulate fluid flow and optimize key geometric parameters (e.g., channel width, constriction length, bypass length) to achieve the desired Q-ratio.
- Fabrication: Fabricate the device in Polydimethylsiloxane (PDMS) using standard soft lithography techniques and a master mold created via photolithography. [20] [26]
2. Experimental Setup:
- Connect the device to a precision syringe pump to control the flow of the cell suspension.
- Use a microscope with time-lapse imaging capabilities to monitor the trapping process and subsequent assays.
3. Cell Loading Procedure:
- Prepare a suspension of your target cells (e.g., HeLa or HEK-293T) at an appropriate concentration to avoid overcrowding.
- Infuse the suspension into the device at a constant, optimized flow rate. The "deterministic" trapping should occur autonomously and sequentially.
Protocol 2: Anti-Clogging via Microbubble Streaming
This protocol describes the integration of a real-time clog prevention system. [1]
1. Device Modification:
- Cavity Design: Incorporate a lateral cavity (e.g., 500µm long, 80µm wide) near the channel constriction prone to clogging.
- Bubble Formation: Upon liquid infusion, an air pocket will be trapped in this cavity, forming a quasi-cylindrical microbubble.
- Transducer Attachment: Affix a piezotransducer to the microchip, aligned to activate the microbubble.
2. System Operation:
- Choose an operational mode:
- Event-Triggered: Use a control system to activate the transducer only when a clog is detected.
- Periodic: Activate the transducer at set time intervals.
- Continuous: For severe clogging risks, run the transducer continuously.
- Activate the piezotransducer, typically near the resonant frequency of the microbubble, to induce strong 3D microstreaming vortices that disrupt particle clusters and prevent arch formation.
The table below summarizes key performance metrics from cited literature to serve as benchmarks for your own experiments.
| Trap Geometry | Cell Type | Trap Sites | Single-Cell Efficiency | Trapping Time / Speed | Key Advantage |
|---|---|---|---|---|---|
| Concatenated T-Junction Pairs [20] | HeLa, HEK-293T | 400 | 90% | < 10 min for 400 sites | High spatial efficiency, deterministic loading |
| V-Shaped Pockets [26] | Yeast (S. cerevisiae) | 624 | High (Precise metric not given) | Not Specified | High-throughput, suitable for long-term assays |
| U-Shaped Posts [27] | Mammalian Cells | Varies | High for single cells | Not Specified | Good for single-cell culture and dynamic perfusion |
| Microbubble Streaming (Anti-Clogging) [1] | Polystyrene Particles | N/A | N/A | Effective real-time prevention | Prevents blockages without moving parts, biocompatible |
Research Reagent Solutions
The table below lists essential materials and their functions for experiments involving hydrodynamic traps.
| Item | Function / Application |
|---|---|
| Polydimethylsiloxane (PDMS) | The most common elastomer for rapid prototyping of microfluidic devices via soft lithography. It is transparent, gas-permeable, and biocompatible. [26] |
| SU-8 Photoresist | A negative, epoxy-based photoresist used to create high-aspect-ratio master molds on silicon wafers during the soft lithography process. [26] |
| Fluorescent Polystyrene Particles | Used for device testing, calibration, and clogging studies. Their size, surface charge, and fluorescence can be tailored to mimic cells. [1] |
| Concanavalin A | A lectin protein used to chemically coat PDMS surfaces to promote cell adhesion and immobilization after hydrodynamic trapping. [26] |
System Workflow and Signaling Diagram
The following diagram illustrates the logical decision process of a deterministic hydrodynamic trapping system and its integration with an anti-clogging mechanism.
Diagram 1: Workflow of a high-efficiency trapping system with integrated anti-clogging.
In microfluidic single-cell analysis, channel clogging is a critical failure point that compromises data integrity, halts experiments, and increases operational costs. Clogging occurs when particles, including biological cells, form blockages at channel constrictions. These blockages can arise from stable static particle arches, single-particle adhesion to channel walls, or particle aggregation [1]. Active microfluidics, which implements external fields such as acoustic waves and tailored fluid dynamics, provides a powerful strategy to overcome these challenges. This technical support center offers targeted troubleshooting guides and protocols to help researchers achieve clog-free manipulation for robust and continuous experimentation.
Troubleshooting Guides & FAQs
FAQ: What are the primary active methods for preventing clogs?
Several active methods have been developed to prevent and mitigate clogging:
- Acoustic Microbubble Streaming: Utilizes piezotransducer-activated microbubbles to generate strong, 3D micro-vortices that disrupt particle arches and clusters near constrictions [1].
- Pulsatile Flow: Involves superimposing oscillating pressure waveforms onto the main flow to periodically dislodge particles before they form stable clogs [2].
- Mechanical Lateral Sieving: Applies low-frequency oscillation directly to the fluid flow to release aggregated particles trapped in filters, enabling continuous operation [9].
Troubleshooting Scenario 1: Clogging at Channel Constrictions
- Problem: Rapid clogging occurs at a narrow constriction used for cell focusing, halting the experiment within minutes.
- Investigation: Check the neck-to-particle size ratio (W/d). Clogging is more probable when this ratio is small (e.g., 1.5) [1].
- Solution: Implement an acoustic microbubble streaming system.
- Design: Integrate a lateral cavity (e.g., width w = 80 µm, length h = 500 µm) adjacent to the constriction to host a quasi-cylindrical microbubble [1].
- Actuation: Affix a piezotransducer to the microchip and actuate it near the bubble's resonant frequency.
- Operation: Run the system in event-triggered mode (activating upon a pressure spike) or periodic mode to prevent clogging without continuous energy input [1].
Troubleshooting Scenario 2: Clogging in a Parallel Filtration Array
- Problem: A multi-channel filtration device with 800 potential clog sites experiences progressively reduced flow and eventual complete blockage [2].
- Investigation: Monitor the flow rate; a steady decline indicates ongoing particle deposition and filter cake formation.
- Solution: Apply a controlled pulsatile flow.
- Setup: Use a high-precision pressure controller (e.g., OB1 MK3+) to generate a sinusoidal pressure input. A Coriolis flow sensor can monitor flow rate reductions correlated with clogging [2].
- Parameters: For a system with an average driving pressure of 150 mbar, introducing a pulsatile amplitude of 50% of the average pressure (75 mbar) at a frequency of 0.1 Hz can nearly double the filter's half-life [2].
- Critical Note: Avoid flow reversal (where pulsatile pressure exceeds average pressure), as it can resuspend particles and accelerate clogging in adjacent channels [2].
FAQ: Can these methods be used with biological cells?
Yes, these methods are designed for biocompatibility. Acoustic microbubble streaming is recognized for its inherent biocompatibility [1]. The lateral flow μ-sieving technique has been successfully demonstrated for the continuous separation of cancer cells (MDA-MB-231) from whole blood without filter clogging [9].
Experimental Protocols for Clog Mitigation
Protocol 1: Implementing Acoustic Microbubble Streaming
This protocol details the setup for preventing clogs at a single constriction using 3D microbubble streaming [1].
- Objective: Real-time prevention of clogging incidents in a microchannel with a constriction.
Materials:
- PDMS microchip with an integrated lateral cavity near the constriction.
- Piezoelectric transducer.
- Function generator and amplifier.
- Pressure-driven flow control system.
- Fluorescent polystyrene particles (e.g., 50 µm or 100 µm) for testing.
Procedure:
- Chip Priming: Rapidly infuse the particle suspension into the microchannel to trap an air bubble within the lateral cavity, forming a microbubble.
- System Setup: Affix the piezotransducer to the microchip and connect it to the function generator and amplifier.
- Frequency Calibration: Apply an acoustic signal sweep to identify the resonant frequency of the microbubble, which produces the strongest microstreaming vortices. This often occurs at frequencies between hundreds of Hz to a few kHz [1].
- Operation:
- Continuous Mode: Activate the transducer continuously for applications requiring constant agitation.
- Event-Triggered Mode: Integrate with a pressure sensor to activate the transducer only when a clog-induced pressure increase is detected.
- Periodic Mode: Program the transducer to activate in short, regular intervals to conserve energy.
- Validation: Use high-speed microscopy to observe the formation of counter-rotating vortices and the subsequent prevention of arch formation at the constriction.
Protocol 2: Establishing Pulsatile Flow for Clog Delay
This protocol outlines the procedure for generating a pulsatile flow to delay clogging in a multi-channel array [2].
- Objective: Significantly extend the operational lifetime of a parallel microchannel filtration device.
Materials:
- Microfluidic chip with parallel channels.
- Precision pressure controller (e.g., Elveflow OB1 MK3+).
- Coriolis flow sensor (e.g., Bronkhorst).
- Data acquisition system for flow rate and video recording.
Procedure:
- Setup Configuration: Connect the pressure controller to the chip inlet and the flow sensor in series at the outlet. Pressurize both inlet and outlet reservoirs to ~150 mbar to limit bubble formation [2].
- Baseline Measurement: Under steady flow conditions, inject the particle suspension and record the time until the flow rate reduces to half its initial value (filter half-life).
- Pulsatile Flow Application:
- Program the pressure controller to output a sinusoidal waveform.
- Set the average pressure to your operating pressure (e.g., 150 mbar).
- Set the amplitude to 50% of the average pressure.
- Set the frequency to 0.1 Hz.
- Monitoring & Optimization: Simultaneously record the flow rate and video footage. Correlate flow rate reductions with visual clogging events. Optimize amplitude and frequency for your specific particle system, as the ideal parameters are system-dependent [2].
Data Presentation: Quantitative Comparisons
The following tables summarize key quantitative data from the cited research to aid in experimental planning and comparison.
Table 1: Performance Comparison of Active Anti-Clogging Techniques
| Method | Reported Efficiency / Improvement | Key Operational Parameters | Application Context |
|---|---|---|---|
| Acoustic Microbubble Streaming [1] | Effective real-time prevention of arch formation and cluster disintegration. | Resonant frequency actuation; Operational modes: Continuous, Periodic, Event-triggered. | Clogging at channel constrictions; biocompatible operations. |
| Pulsatile Flow [2] | ~100% improvement in filter half-life compared to steady flow. | Amplitude: 50% of average pressure; Frequency: 0.1 Hz (system-dependent). | Parallel microchannel arrays; filtration systems. |
| Lateral Flow μ-Sieving [9] | 100% separation efficiency; >98% retrieval rate of target particles. | Low-frequency oscillation (70-230 Hz tested). | Continuous size-based separation of microparticles and cancer cells from blood. |
Table 2: Reagent and Material Solutions for Featured Experiments
| Research Reagent / Material | Function / Explanation |
|---|---|
| Polystyrene Fluorescent Particles [1] | Model particles for testing and validating anti-clogging device performance. Sizes of 50 µm and 100 µm are common. |
| Glycerol Aqueous Solution (23.8 w-w%) [1] | Used to achieve neutral buoyancy for particles, matching their density to the surrounding medium to minimize sedimentation. |
| PDMS (Polydimethylsiloxane) [9] [20] | A transparent, biocompatible polymer used for rapid prototyping of microfluidic devices via soft lithography. |
| SU-8 Photoresist [9] | A high-contrast, negative-tone photoresist used to create master molds for microfluidic channel fabrication. |
Workflow Visualization
The following diagram illustrates the logical decision pathway for selecting and implementing an appropriate anti-clogging strategy based on the experimental problem.
Anti-Clogging Strategy Decision Guide
Fundamental Concepts: Why Do Microfluidic Channels Block?
Microfluidic channel blockage, or fouling, is a transient process that begins the moment a fluid comes into contact with a surface. It involves the undesired adhesion and accumulation of biological or chemical substances, leading to clogging, increased flow resistance, and compromised experimental results [28].
The Mechanisms of Contaminant Adhesion
Fouling occurs through a complex interplay of physical and chemical interactions between contaminants and the channel surface [29] [28].
- Chemical Interactions include the formation of covalent bonds, ionic bonds, and coordination bonds. For instance, proteins rich in cysteine can bind to gold surfaces via stable gold-thiol bonds, while amino groups on microorganisms can form Schiff bases with aldehyde groups on a surface [29].
- Physical Adsorption involves hydrogen bonds, van der Waals forces, and hydrophobic interactions. Lipids and other organic compounds can displace interfacial water via hydrophobic interactions and then bind through van der Waals forces, forming a persistent film [29].
- Mechanical Interlocking occurs when particles or macromolecules in the fluid, such as proteins or colloidal substances, become physically adsorbed and deposited within or on the pores of a surface, leading to blockage. In membrane-based systems, this is a primary cause of reduced permeate flux [29].
The Spatiotemporal Process of Fouling
The adhesion process typically follows a temporal sequence with distinct spatial characteristics [29] [28]:
- Conditioning Layer Formation: The surface is initially coated by a layer of organic macromolecules (e.g., proteins or polysaccharides).
- Microorganism Adhesion: This conditioning layer provides a favorable microenvironment for the subsequent adhesion of bacteria or cells.
- Biofilm Development: Adhered microorganisms secrete metabolic products, such as extracellular polymeric substances (EPS), which further alter the surface morphology and chemistry, promoting further contamination and eventual channel blockage.
The diagram below illustrates this progressive fouling mechanism.
Troubleshooting Guide: Diagnosis and Resolution of Blockage Issues
Use the following tables to diagnose your specific issue and identify potential solutions.
Table 1: Diagnosing Fouling Types and Causes
| Observed Symptom | Potential Contaminant(s) | Primary Adhesion Mechanism(s) | Common Experiment Types |
|---|---|---|---|
| Gradual, consistent pressure increase | Proteins, Polysaccharides | Hydrophobic interactions, Hydrogen bonding, Covalent bonding | Single-cell proteomics, Serum culture |
| Rapid pressure spike or complete clogging | Cells, Cell debris, Aggregates | Mechanical interlocking, Physical adsorption | Whole blood analysis, Tissue homogenate processing |
| Slow, steady decline in flow rate; surface feels slimy | Bacteria, Microorganisms, Biofilms | Secretion of adhesive EPS; Chemical & physical bonding | Long-term cell culture, Perfusion experiments |
Table 2: Selecting Antifouling Strategies Based on Mechanism
| Antifouling Mechanism | Key Principle | Ideal Application | Coating Example(s) |
|---|---|---|---|
| Adjust Surface Energy | Modify surface free energy to a range (20-30 mN/m) that minimizes contaminant adhesion [29]. | Broad-spectrum prevention against proteins and cells. | Protein-resistant polymers (e.g., PEG). |
| Create Superhydrophilic Surfaces | Form a tightly bound water layer that acts as a physical and energetic barrier to foulants [29]. | High-salinity buffers, protein solutions. | Zwitterionic polymers, PEG-based hydrogels. |
| Create Superhydrophobic/Superslippery Surfaces | Prevent water adhesion and enable foulants to be easily shed [29] [30]. | Environments with particulate matter or oil. | Fluorinated coatings, Slippery Liquid-Infused Porous Surfaces (SLIPS). |
| Incorporate Biocidal/Bioactive Agents | Actively kill adhering microorganisms to prevent biofilm formation [30]. | Long-term experiments, bacterial cultures. | Copper-based coatings, photo-responsive coatings (e.g., ZnO, TiO₂) [30]. |
Frequently Asked Questions (FAQs)
Q1: My single-cell analysis device consistently clogs when processing whole blood samples. What is the fastest way to mitigate this? The most immediate mitigation is a combination of surface pretreatment and operational adjustment.
- Surface Pretreatment: Prior to your experiment, passivate the microchannels by flushing with a 1-5% w/v solution of a passivation protein like bovine serum albumin (BSA). This protein will adsorb to the surface, blocking sites that would otherwise bind blood components.
- Operational Adjustment: Incorporate a pre-filtering step using an in-line membrane or a dedicated chip region with larger features (e.g., >20 µm) to remove large aggregates and cell debris before the sample reaches the sensitive analytical sections [28].
Q2: For long-term cell culture studies, how can I prevent biofilm formation without affecting cell viability? Utilize non-biocidal, fouling-release coatings. These do not kill cells but create a surface that makes it difficult for them to remain attached.
- Coating Strategy: Apply a grafted layer of Poly(ethylene glycol) (PEG) or a zwitterionic polymer to the channel surfaces not used for intended cell adhesion. These coatings create a highly hydrophilic, hydration layer that resists protein adsorption and subsequent cell attachment [29].
- Material Choice: If possible, fabricate your device from a fouling-release material like a silicone elastomer (e.g., PDMS). Its low surface energy makes it harder for biofilms to form strong adhesion.
Q3: Are "green" or bio-based coatings effective for sensitive analytical applications? Yes, antifouling bio-coatings are a rapidly advancing field. They are composed of biodegradable and non-toxic biopolymers (e.g., polysaccharides, proteins) and can integrate multiple functions like antibacterial and antiviral properties. Their non-toxic nature makes them particularly suitable for human-related fields, including medical devices and diagnostic microfluidics, where analyte integrity is paramount [29].
Q4: I observe inconsistent results between device runs. Could minor surface fouling be the cause? Absolutely. Inconsistent fouling can lead to:
- Variable adsorption of analytes, reducing signal and reproducibility.
- Uncontrolled flow resistance, altering shear stresses on cells and delivery timing.
- Non-specific binding, creating high background noise. Implementing a rigorous and standardized channel cleaning and surface regeneration protocol (see Section 4.1) between each run is critical for achieving high-quality, reproducible data.
Experimental Protocols for Validation and Maintenance
Protocol: Surface Passivation with PEG-Silane
Function: Creates a dense, hydrophilic brush layer that minimizes non-specific adsorption of proteins and cells.
Workflow Diagram:
Detailed Methodology:
- Surface Activation: Place your glass or silicon chip in a plasma cleaner. Treat it with oxygen plasma at 100 W for 1-2 minutes. This step creates reactive hydroxyl (-OH) groups on the surface.
- Silanization: Immediately prepare a 2-5 mM solution of methoxy-PEG-silane (e.g., mPEG-silane, MW 2000-5000) in anhydrous toluene. Transfer the activated chip into this solution and incubate at room temperature for 1 hour under an inert atmosphere (e.g., nitrogen or argon glovebox) to prevent hydrolysis.
- Curing: Remove the chip from the solution and cure it in an oven at 70-80 °C for 2-4 hours to complete the covalent grafting.
- Rinsing and Storage: Thoroughly rinse the functionalized chip with fresh toluene, ethanol, and finally deionized water to remove any physisorbed molecules. The chip can be stored dry or in phosphate-buffered saline (PBS) for immediate use. Confirm modification by an increase in water contact angle.
Protocol: Evaluating Coating Efficiency with a Protein Fouling Test
Function: Quantitatively assesses the antifouling performance of your modified surface against a standard protein.
Workflow Diagram:
Detailed Methodology:
- Preparation: Prepare a 1 mg/mL solution of a fluorescently-labeled protein, such as FITC-Bovine Serum Albumin (FITC-BSA), in PBS.
- Exposure: Perfuse this solution through your modified microchannel at a defined shear rate for a set period (e.g., 30 minutes at a shear rate of 100 s⁻¹).
- Rinsing: Thoroughly flush the channel with pure PBS buffer for at least 10 minutes to remove any unbound protein.
- Quantification: Use fluorescence microscopy to image the channel surface. Quantify the adsorbed protein by measuring the average fluorescence intensity per unit area. Compare this value to the intensity on an uncoated control channel to calculate the percentage reduction in fouling. A high-performance coating should achieve >90% reduction.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Antifouling Surface Modifications
| Reagent / Material | Function / Role in Antifouling | Example Application |
|---|---|---|
| Poly(ethylene glycol) (PEG)-Silane | Forms a hydrophilic, steric barrier that reduces protein adsorption and cell adhesion. | Covalent grafting to glass/silicon surfaces for passivation. |
| Zwitterionic Monomers (e.g., SBMA, CBMA) | Creates a superhydrophilic surface that binds a strong hydration layer via electrostatic interactions. | Polymer brushes for ultra-low fouling surfaces in complex media. |
| Photo-responsive Polymers (e.g., with Azobenzene, Coumarin) | Enables light-controlled release of foulants or activation of antifouling agents [30]. | "Self-cleaning" coatings that shed fouling layers upon light exposure. |
| Titanium Dioxide (TiO₂) / Zinc Oxide (ZnO) Nanoparticles | Provides photocatalytic activity; generates reactive oxygen species (ROS) under UV light to degrade organic foulants [30]. | Composite coatings for active, biocidal antifouling. |
| Bovine Serum Albumin (BSA) | Acts as a passivation agent by adsorbing to surfaces and blocking sites for further protein binding. | Quick, non-covalent surface passivation for protein assays. |
Troubleshooting Guides
Common Clogging Issues and Solutions in Microfluidic Systems
The following table summarizes frequent problems encountered during sample preparation that lead to channel blockages and the corresponding corrective actions.
| Problem | Possible Causes | Recommended Solutions |
|---|---|---|
| Clogging from particle aggregates [1] [31] | - Particle accumulation at constrictions- Formation of stable particle arches- High particle concentration in sample | - Implement pre-filtration to remove large aggregates [32]- Use continuous or periodic microbubble streaming near constrictions to disrupt arch formation [1]- Optimize sample concentration and introduce surface charge (e.g., sulfate groups) to prevent agglomeration [1] |
| Low cell yield / Low viability [33] | - Over- or under-dissociation of tissue- Cellular damage from enzymatic treatment | - Switch to a less digestive enzyme type (e.g., from trypsin to collagenase)- Reduce enzyme concentration or incubation time- Add bovine serum albumin (BSA) (0.1-0.5% w/v) to dilute proteolytic action [33] |
| High cell yield / Low viability [33] | - Enzyme is overly digestive or concentration is too high | - Reduce enzyme concentration and/or incubation time- Add enzyme inhibitors like soybean trypsin inhibitor (0.01-0.1% w/v) [33] |
| Clogging from cell clumps [34] | - Cell clumping after fixation steps (e.g., methanol addition) | - Dispense ice-cold methanol slowly (>30 seconds) during fixation to prevent clump formation [34]- Perform additional pipette mixing to dissociate visible clumps before loading the chip [34] |
| Mechanical blockages [31] | - Inadequate microchannel design- Poor surface properties leading to particle adhesion- Material incompatibility causing degradation | - Perform careful design iterations and simulations during device development [31]- Optimize channel surface wettability to control fluid behavior [31]- Ensure material compatibility with all reagents and samples [31] |
Pre-filtration Selection Guide
Selecting the correct pre-filtration method is critical for removing particulates without losing your target cells. The table below compares key techniques.
| Filtration Technique | Typical Pore Size / Purpose | Key Applications & Notes |
|---|---|---|
| Prefiltration [32] | Broad range of retention ratings | Removes high contaminant content; protects downstream membrane filters [32]. |
| Membrane Filtration [32] | 0.025 µm - 10 µm [35] | Clarification and sterilization; prone to clogging; used to polish fluids [32]. |
| Crossflow Filtration [32] | Micro- or Ultrafiltration | Fluid sweeps over membrane to prevent blocking; allows for sample concentration [32]. |
| Syringe Filtration [32] [35] | 0.1 µm - 0.22 µm common | Removes particles, bacteria for UPLC, UHPLC, HPLC; ensures sample integrity [32]. For microfluidic sample prep, use 0.1µm membranes to remove mycoplasma [36]. |
| Centrifugal Filters [32] | Various MWCO / pore sizes | Rapid separation and concentration using centrifugal force; ideal for sample clarification [32]. |
Frequently Asked Questions (FAQs)
Q1: How can I prevent clogging in my microfluidic device without using complex external systems?
Consider integrating an acoustic microbubble streaming strategy directly into your chip design [1]. This involves creating a lateral cavity near channel constrictions to trap a microbubble. When activated by a piezotransducer, the bubble generates strong, 3D microstreaming vortices that disrupt particle arches and break up clusters in real-time [1]. This method is biocompatible and can be operated in event-triggered, continuous, or periodic modes to suit your application.
Q2: My samples have low cell viability after dissociation, which leads to clumping and clogging. How can I optimize this?
Achieving a balance between high cell yield and high viability is key [33]. If you have low yield and low viability, you are likely over-dissociating the tissue. Switch to a less digestive enzyme (e.g., from Trypsin to a blend like Collagenase) and/or decrease the working concentration [33]. If you have high yield but low viability, the enzyme is too harsh; reduce the concentration or incubation time, or add a protective agent like Bovine Serum Albumin (BSA) at 0.1-0.5% (w/v) to the dissociation mixture [33].
Q3: Why is pre-filtration essential before injecting a sample into a microfluidic system, and what filter should I use?
Pre-filtration is crucial because particles and cell clumps can obstruct microchannels, which are often only tens to hundreds of microns wide [31] [6]. This prevents clogging in sensitive areas like constrictions and ensures uninterrupted flow and analysis. For most applications, sterile syringe filters with a 0.1µm membrane are recommended. This pore size is small enough to remove troublesome mycoplasma contamination, which can pass through standard 0.22µm filters [36].
Q4: I am losing many cells during the washing and centrifugation steps of my protocol. What could be wrong?
A common error is centrifuging at incorrect speeds. Ensure you are using the correct relative centrifugal force (RCF), measured in g-force, not RPM [34]. Centrifuging at lower than recommended speeds will fail to pellet the cells, resulting in loss when the supernatant is discarded. Furthermore, when discarding the supernatant, always ensure the cell pellet remains submerged in a small residual volume of fluid (e.g., ~40 µL) to avoid accidentally discarding cells [34].
Research Reagent Solutions
The following table lists essential materials and reagents used in sample preparation for microfluidic analysis.
| Item | Function / Application |
|---|---|
| Syringe Filters (0.1 µm) [36] | Final pre-filtration to remove particulates and sub-micron contaminants like mycoplasma prior to sample loading. |
| Collagenase/Trypsin Enzymes [33] | For tissue dissociation to isolate individual cells; selection depends on tissue type and required digestive strength. |
| Bovine Serum Albumin (BSA) [33] | Used as an additive (0.1-0.5% w/v) in dissociation mixtures to protect cells from overly proteolytic action. |
| Polystyrene Microspheres [1] | Often used as model particles for testing and optimizing anti-clogging strategies in microchannels. |
| Microbubble Cavity-Integrated Chip [1] | A specialized microfluidic chip with designed cavities to trap microbubbles for acoustic streaming-based clog disruption. |
| Soybean Trypsin Inhibitor [33] | An enzyme inhibitor (used at 0.01-0.1% w/v) to quickly halt trypsin activity and prevent over-digestion of cells. |
Workflow for Clog Prevention
The diagram below outlines a logical workflow for preventing channel blockage, integrating sample preparation and on-chip solutions.
Practical Protocols for Clog Prevention, Detection, and System Recovery
Troubleshooting Guides
Frequently Asked Questions (FAQs)
Q1: What are the primary indicators of a partial clog in my microfluidic channel?
A partial clog is often signaled by a gradual increase in the upstream pressure, a decrease in the observed flow rate at the outlet, and an increase in pressure fluctuations or instability. In single-cell analysis devices, this may also manifest as a inconsistent cell arrival times at a detection or trapping zone [37].
Q2: How can I differentiate between a pressure spike caused by a clog and one caused by an air bubble?
Air bubbles often cause sharp, transient pressure spikes that may resolve quickly as the bubble passes through or dissolves. A true clog typically causes a sustained increase in baseline pressure. Monitoring the pressure trace over time is key: a clog shows a step-change, while a bubble shows a brief peak [37] [38].
Q3: My pressure data is very noisy. Is this a sign of clogging, or could it be another issue?
Noisy pressure data can indicate several issues. While unstable clog formation can cause fluctuations, it can also be a sign of:
- Air bubbles in the system: Small bubbles passing through the flow cell can cause high-frequency noise [38].
- Unstable flow source: Check that your pressure- or syringe-pump is functioning correctly.
- Incorrect sensor placement: As noted in production data analysis, ensuring the pressure gauge is in the correct location (e.g., upstream of the constriction) is critical for valid data [39].
- Liquid loading/slugging: In systems with multiple phases, unstable flow can create pressure noise, analogous to liquid loading in wellbores [39].
Q4: What is an event-triggered anti-clogging activation mode?
This is a control strategy where a monitoring system (e.g., a pressure sensor) detects a predefined signature of clog formation, such as a pressure threshold being exceeded. Upon detection, it automatically triggers a counter-measure, such as activating a microbubble to disrupt the clog, before returning to normal operation. This is more efficient than continuous activation [1].
Troubleshooting Common Problems
The table below summarizes common issues, their possible causes, and recommended actions.
| Problem | Possible Causes | Recommended Diagnostic & Action |
|---|---|---|
| Sudden, sustained pressure increase | Full channel blockage by a large cell cluster or debris. | Diagnostic: Check if pressure remains high after flow is stopped and restarted. Action: Initiate a flow reversal or pulse if available [1]. Activate an integrated anti-clogging mechanism (e.g., acoustic microbubble) [1]. |
| Gradual pressure creep with high fluctuation | Progressive clog formation and unstable arching of particles near a constriction [1]. | Diagnostic: Observe correlation between particle concentration and pressure rise rate. Action: Switch to periodic or event-triggered anti-clogging mode to disrupt arches before they stabilize [1]. |
| Low or no flow rate, but pressure is normal | Syringe pump plunger slip, loose tubing connections, or an empty sample reservoir [38]. | Diagnostic: Visually inspect all fluidic connections and reservoir volumes. Action: Tighten connections, refill reservoir, and ensure the syringe pump is properly engaged. |
| High background noise in data | Electronic noise from the sensor or air bubbles in the flow cell [38]. | Diagnostic: Run the system with pure buffer to establish a baseline. Action: Prime the system thoroughly to remove bubbles. Check instrument ground connections and ensure photomultiplier tube (PMT) voltages are not set excessively high [38]. |
Experimental Protocols & Data Presentation
Protocol: Implementing Microbubble Streaming for Clog Prevention
This protocol is adapted from research detailing the use of 3D microbubble streaming as a dynamic anti-clogging solution [1].
1. Objective: To integrate and activate an acoustic microbubble system for real-time clog prevention and disruption in a microfluidic channel.
2. Materials:
- Microfluidic Chip: Fabricated with a straight channel (e.g., 450 µm wide) containing a constriction (e.g., 150 µm wide) and a lateral cavity (e.g., 500 µm x 80 µm) for microbubble formation adjacent to the constriction [1].
- Piezoelectric Transducer (Piezotransducer): Affixed to the microchip near the bubble cavity.
- Function Generator & Amplifier: To drive the piezotransducer at the required frequency and amplitude.
- Pressure Sensors: Integrated upstream and/or downstream of the constriction for feedback.
- Control System & Software: Capable of operating in continuous, periodic, or event-triggered mode.
- Sample: Cell or particle suspension (e.g., 50 µm or 100 µm polystyrene particles) prepared in a neutrally buoyant aqueous solution (e.g., with 23.8% w-w glycerol) to prevent sedimentation [1].
3. Methodology:
- Priming and Bubble Trapping: Completely prime the microchannel with your sample fluid. Rapidly infuse liquid to trap a gas pocket (e.g., air) within the lateral cavity, forming a quasi-cylindrical microbubble [1].
- Calibration and Frequency Sweep: Activate the piezotransducer and perform a frequency sweep to identify the resonant frequency of the microbubble, which generates the strongest 3D counter-rotating microvortices [1].
- System Integration: Connect the pressure sensor output to the control system. Define the triggering parameters (e.g., if pressure > X kPa for Y seconds, activate piezotransducer at Z frequency and amplitude for A seconds).
- Operation: Initiate your single-cell experiment. The system can now operate in:
- Continuous Mode: Piezotransducer is always on. Simple but may affect other processes.
- Periodic Mode: Piezotransducer activates at fixed intervals.
- Event-Triggered Mode (Recommended): The system monitors pressure in real-time and only activates the anti-clogging mechanism when a clog is imminent [1].
The table below summarizes key parameters from the referenced anti-clogging study using microbubble streaming [1].
| Parameter | Value / Range | Experimental Context |
|---|---|---|
| Constriction Width (W) | 150 µm | Square cross-section for neck formation. |
| Particle Sizes (d) | 50 µm, 100 µm | Polystyrene fluorescent particles. |
| Neck-to-Particle Ratio (W/d) | 3, 1.5 | Tests clogging propensity with different size ratios. |
| Bubble Cavity Dimensions | 500 µm (length) x 80 µm (width) | Optimized to prevent bubble detachment or weak streaming [1]. |
| Actuation Mode | Event-triggered, Continuous, Periodic | Provides flexibility for different application needs [1]. |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in the Context of Clog Prevention |
|---|---|
| Polystyrene Microspheres | Used as model cells (e.g., 50-100 µm) to simulate biological cells and study clogging dynamics at constrictions in a controlled manner [1]. |
| Glycerol Solution (23.8% w-w) | Creates a neutrally buoyant medium for particles/cells, minimizing sedimentation that could contribute to clogging and ensuring consistent particle entry into constrictions [1]. |
| Negatively Charged Surfactants | Sulfate groups on microspheres provide surface charge, preventing agglomeration and adhesion to channel walls (often made of PDMS), thereby reducing one mechanism of clogging [1]. |
| Piezoelectric Transducer | The actuator that converts electrical signals into mechanical vibrations, which drive the oscillation of the microbubble to generate powerful, localized microstreaming flows [1]. |
| Fixable Viability Dyes | Allows for the identification and gating of dead cells during flow experiments, which is important as dead cells are more prone to aggregation and can initiate clogs [38]. |
Diagnostic and Anti-Clogging Workflows
Pressure Anomaly Diagnostic Logic
Microbubble Anti-Clogging Mechanism
Preventive Maintenance Schedules and Cleaning Regimens
Preventing channel blockage is a critical challenge in microfluidic single-cell analysis research. Clogging can disrupt experiments, damage delicate cellular samples, and compromise data integrity. This technical support center provides targeted troubleshooting guides and FAQs to help researchers maintain optimal device performance and ensure the reliability of their experimental results.
Understanding Clogging in Microfluidic Systems
Why is clogging a particular problem for single-cell analysis?
Microfluidic devices for single-cell analysis feature channel dimensions and constrictions designed to manipulate and trap individual cells. These small features are highly susceptible to blockage from cell clusters, debris, or aggregates [1]. Clogging is especially detrimental in long-term cultivation experiments, where it can halt nutrient supply and waste removal, leading to cell death and experiment failure [40].
What are the common causes of clogging?
The primary causes include:
- Particle Aggregates: Cells or other particles can form clusters that are too large to pass through channel constrictions [1].
- Filter Cake Formation: Particles can accumulate at constrictions, forming an "arch" or "filter cake" that blocks the flow for other particles [2].
- Inappropriate Channel Design: If supply channels are not wide enough to accommodate the cells being used, clogging is likely to occur during the loading process [40].
Anti-Clogging Techniques and Methodologies
Several advanced methods have been developed to mitigate or resolve clogging in microfluidic channels. The following table summarizes the key approaches.
Table 1: Comparison of Active Anti-Clogging Techniques
| Technique | Principle | Key Parameters | Best Use Cases |
|---|---|---|---|
| Microbubble Streaming [1] | Uses acoustic actuation of a microbubble to generate 3D counter-rotating vortices (microstreaming) that break apart clusters and prevent arch formation. | Actuation frequency and amplitude; bubble size and position. | Real-time, biocompatible prevention in continuous-flow systems; effective near constrictions. |
| Pulsatile Flow [2] | Applies a oscillating (pulsatile) pressure to the flow, creating high shear conditions that can erode particle aggregates and rearrange filter cakes. | Pulsation amplitude and frequency; avoiding flow reversal in multi-channel arrays. | Delaying clogging in parallel microchannel arrays; can nearly double filter half-life. |
| Flow Reversal [2] | Periodically reverses the direction of flow to resuspend particles that have begun to accumulate. | Reversal timing and duration. | Systems where temporary backflow does not harm the sample or experiment. |
Experimental Protocol: Mitigating Clogging with Microbubble Streaming
This protocol is adapted from recent research demonstrating real-time clog prevention [1].
1. Objective: To prevent clogging and disintegrate particle clusters at a channel constriction using 3D microbubble streaming. 2. Materials: * PDMS-based microfluidic device with an integrated lateral cavity for microbubble formation near the constriction [1]. * Piezoelectric transducer affixed to the microchip. * Function generator to drive the transducer. * Polystyrene microparticles or cell suspension in a neutrally buoyant aqueous solution (e.g., with glycerol) [1]. * Syringe pump or pressure-driven flow control system. * Microscope for visualization. 3. Methodology: * Priming: Rapidly infuse the liquid sample into the microchannel, trapping an air pocket in the lateral cavity to form a quasi-cylindrical microbubble [1]. * Activation: Activate the piezotransducer with a sinusoidal signal. The frequency should be near the resonant frequency of the microbubble to induce strong oscillatory motion. * Operation: The bubble oscillation interacts with the channel walls, generating a steady secondary flow (microstreaming) with strong shear stresses. This flow disrupts particle arches and disintegrates clusters before they can cause a permanent clog. * Control Modes: The system can be operated in: * Continuous Mode: For constant protection in high-risk applications. * Periodic Mode: To conserve energy and minimize potential effects on cells. * Event-Triggered Mode: Where a detected increase in fluidic resistance activates the bubble to clear a nascent clog [1].
Experimental Protocol: Delaying Clogging with Pulsatile Flow
This protocol is based on a study investigating clogging dynamics in microfluidic arrays [2].
1. Objective: To establish pulsatile flow parameters that significantly delay clogging in a parallel microchannel array. 2. Materials: * PDMS microfluidic chip with multiple parallel channels, each containing constrictions. * Pressure controller (e.g., Elveflow OB1 MK3+) capable of generating precise sinusoidal pressure profiles. * In-line flow sensor (e.g., Bronkhorst Coriolis) to monitor flow rate and detect clogging. * Particle suspension (e.g., 2 µm polystyrene beads). 3. Methodology: * Setup: Pressurize both inlet and outlet reservoirs to improve gas solubility and limit bubble formation [2]. * Calibration: Under steady flow (e.g., 150 mbar), establish a baseline flow rate and average clogging rate for your system. * Apply Pulsations: Switch the pressure controller to a pulsatile mode. A recommended starting point is an amplitude of 50% of the average pressure and a frequency of 0.1 Hz [2]. * Optimization: Systematically test different amplitude and frequency combinations. Use the flow sensor and visual observation to determine the settings that maximize the operational life of the device. Avoid amplitudes that cause full flow reversal, as this can accelerate clogging in multi-channel systems by transferring particles to adjacent channels [2]. * Validation: Compare the total processed volume and "filter half-life" between steady and optimized pulsatile flow conditions.
Preventive Maintenance and Cleaning
A proactive approach is the most effective strategy for managing clogging.
How can I prevent my microfluidic devices from clogging?
Pre-Experiment Best Practices:
- Filter All Fluids: Pass all buffers, media, and cell suspensions through an appropriate filter (e.g., 0.2 µm or smaller) before introducing them into the microfluidic device to remove pre-existing aggregates and particulates [41].
- Ensure Neutral Buoyancy: For particle suspensions, adjust the density of the carrier fluid (e.g., with glycerol) to prevent sedimentation, which can lead to local aggregation and clogging [1].
- Design Against Clogging: During the chip design phase, ensure that supply channels are wide enough for the cells being used and that constrictions are appropriately sized [40].
Operational Best Practices:
- Implement Pulsatile Flow: As described in Section 2.2, integrating weak pulsations into your flow protocol can dramatically extend device lifetime [2].
- Start with a Clean Device: Always use devices that have been manufactured and stored in a clean environment to avoid introducing contaminants from the outset [41].
What is the best way to clean a contaminated microfluidic device?
Cleaning a contaminated device is challenging. The most practical approach is prevention. However, for device reuse:
- Address the Root Cause: Identify and eliminate the source of contamination (e.g., unfiltered fluids, dirty storage conditions) [41].
- Storage: Store microfluidic devices in a clean, enclosed container flushed with HEPA-filtered air to prevent particulate accumulation [41].
- Plasma Cleaning: For PDMS devices, oxygen plasma treatment can be used to clean surfaces before bonding and initial use. However, this is not typically effective for cleaning internal channels post-contamination [41].
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions
| Item | Function | Example & Notes |
|---|---|---|
| Glycerol Solution | Adjusts the density of the carrier fluid to achieve neutral buoyancy for particles or cells, preventing sedimentation and associated clogging [1]. | 23.8% (w/w) glycerol in water for polystyrene particles [1]. Concentration must be optimized for specific cell types. |
| Filtered Buffers & Media | Removes pre-existing particles and aggregates from all fluids before they enter the microfluidic system, which is the first line of defense against clogging [41]. | Use a 0.2 µm syringe filter for most aqueous solutions. For cell-compatible media, ensure the filtering process does not remove essential nutrients. |
| DMSO Cryoprotectant | Used for the cryopreservation of cell lines, ensuring a healthy, viable, and genetically stable cell stock for experiments, which reduces the chance of introducing dead cell debris [42]. | Usually 5–10% in serum-containing media. Can be toxic to some cells; alternatives include glycerol or commercial solutions like Bambanker [42]. |
| Polystyrene Microspheres | Act as model cells or particles for testing and optimizing microfluidic device performance, including anti-clogging techniques, without the variability of biological samples [1]. | Available in various sizes (e.g., 50 µm, 100 µm); often fluorescent for easy visualization. Stabilized with negatively charged sulfate groups to prevent agglomeration [1]. |
Frequently Asked Questions (FAQs)
Q1: My channels keep clogging during long-term cell culture. What can I do? A: For long-term cultivation, ensure your design uses a chamber height that appropriately restricts fluid flow to supply channels, promoting diffusive mass exchange and reducing the risk of chambers being blocked by debris [40]. Additionally, consider integrating a pulsatile flow regime, which has been shown to delay clogging and extend the operational life of devices in continuous systems [2].
Q2: Are there any "passive" anti-clogging methods that don't require external power? A: Yes, passive methods exist, such as designing loops or spiral channels to induce recirculating Dean flows, or using lateral vibration microfluidic sieving. However, the effectiveness of these passive methods compared to active methods like microbubble streaming or controlled pulsatile flow may be limited in complex applications [1].
Q3: How does the choice of chip material impact clogging and cleaning? A: PDMS is the most common material due to its ease of fabrication, optical transparency, and gas permeability [43] [40]. Its flexibility allows for the creation of high-aspect-ratio structures. However, its surface properties can attract certain contaminants. Glass chips offer excellent chemical resistance and mechanical stability but are more complex to fabricate [43]. The best practice for both is to prevent contamination from the start, as cleaning internal channels post-clogging is notoriously difficult [41].
Experimental Workflow for Clog-Free Operation
The following diagram outlines a logical workflow for preventing and addressing clogging in microfluidic single-cell analysis, based on the methodologies discussed.
Optimal Trap Architecture Design Principles from Fluid Dynamics Simulations
Troubleshooting Guides
FAQ 1: How can I prevent clogging in my single-cell trapping device?
Problem: Microchannels become blocked by cell clusters or stable particle arches, disrupting experiments and requiring device replacement [1].
Solutions:
- Active Microbubble Streaming: Integrate a lateral cavity near constrictions to trap a microbubble. When acoustically activated, it generates counter-rotating vortices that disrupt arch formation and break apart clusters [1].
- Passive Geometric Design: Implement deterministic hydrodynamic traps based on the "least flow resistance path" principle. These designs sequentially guide cells to empty traps, reducing the chance of multiple cells blocking a single constriction [20].
- Pulsatile Flow: Use optimized pulsatile flows instead of steady flow, which has been shown to significantly delay microchannel clogging [1].
- Surface Charge Management: Utilize particles/cells stabilized with negatively charged sulfate groups. The resulting repulsion hinders agglomeration and adhesion to channel walls [1].
Recommended Experiment: To test your anti-clogging system, prepare a suspension of polystyrene particles with neutral buoyancy. Monitor the channel under flow conditions with and without the anti-clogging mechanism active. Effective systems will show particles passing constrictions without forming stable blockages [1].
FAQ 2: My single-cell trapping efficiency is low. How can I improve it?
Problem: Cells are not reliably trapped in designated sites, or multiple cells occupy a single trap.
Solutions:
- Optimize Trap Geometry: Use a compact configuration of concatenated T and inverse T junction pairs. This design saves space and increases trapping speed compared to long serpentine channels [20].
- Apply Deterministic Principles: Ensure your design follows the "least flow resistance path" principle. An empty trap should have lower flow resistance than the bypass channel, but this ratio should reverse once a cell is trapped [20].
- Adjust Constriction Size: Design trap constrictions with dimensions slightly smaller than your target cells. For cells with rigid walls, chambers with heights lower than cell diameter can effectively retain them [44].
- Simulate First: Use Computational Fluid Dynamics (CFD) simulation to model particle paths and optimize key geometric parameters before fabrication [20] [45].
Quantitative Targets: A well-optimized device can achieve 100% cell trapping and 90% single-cell trapping efficiency over 400 trap sites [20].
FAQ 3: Bubbles become trapped in my microfluidic device. How do I prevent this?
Problem: Air bubbles become stuck in microchannels, interrupting fluid paths and affecting device performance.
Solutions:
- Modify Surface Properties: Create hydrophilic channel walls with lower contact angles. Simulations show a 90° contact angle allows bubbles to pass through constrictions, while a 22.5° angle causes entrapment [46].
- Design Smooth Transitions: Implement arc contours at channel transitions for smooth fluid movement, reducing areas where bubbles can nucleate [20].
- Degas Materials: Thoroughly degas PDMS before curing and all solutions before introducing them to the microfluidic device [44].
Experimental Protocol: To test bubble formation, use your device with a standardized solution at varying flow rates. Observe common trap sites for bubble entrapment. Treat channels with plasma oxidation to increase hydrophilicity if bubble entrapment persists [46].
Quantitative Data Tables
Table 1: Performance Comparison of Single-Cell Trapping Architectures
| Trap Architecture | Trapping Efficiency | Single-Cell Efficiency | Operation Time | Footprint Savings |
|---|---|---|---|---|
| Concatenated T-junction pairs [20] | 100% | 90% (over 400 traps) | <10 minutes | 2-fold improvement |
| Serpentine channel design [20] | Not specified | Lower than T-junction | >30 minutes | Baseline |
| PDMS pillar traps [45] | High (simulation validated) | Efficient for suspension/adherent cells | Not specified | Compact layout |
Table 2: Anti-Clogging Technique Comparison
| Technique | Mechanism | Best For | Limitations |
|---|---|---|---|
| Microbubble Streaming [1] | 3D counter-rotating vortices disrupt clusters | Continuous systems; long-term operations | Requires integrated actuator |
| Pulsatile Flow [1] | Flow variations prevent stable arch formation | Applications tolerant to flow variations | Less effective than active methods |
| Deterministic Hydrodynamic Trapping [20] | Sequential, ordered trapping prevents overcrowding | High-efficiency single-cell analysis | Requires precise geometric control |
| Dean Flow Fractionation [1] | Curved channels create lateral lift forces | Particle separation by size | Limited application for trapping |
Table 3: Bubble Entrapment Prevention Guidelines
| Parameter | Entrapment Condition | Clear Passage Condition | Recommendation |
|---|---|---|---|
| Wall Contact Angle [46] | 22.5° | 90° | Use hydrophilic coatings |
| Channel Geometry | Sharp transitions | Smooth, arc contours [20] | Design with gradual constrictions |
| Flow Rate | Sudden changes | Gradual ramping [46] | Implement controlled flow acceleration |
Experimental Protocols
Protocol 1: Implementing and Testing Microbubble Anti-Clogging
Purpose: Integrate and validate an active anti-clogging system using microbubble streaming.
Materials:
- PDMS microfluidic device with lateral cavities for microbubble formation
- Piezotransducer and function generator
- Polystyrene particles (50µm and 100µm) with fluorescent labeling
- Syringe pump and tubing
- Microscope with high-speed camera
Methodology:
- Fabrication: Create microchannels with rectangular cross-sections (150µm × 450µm) transitioning to a constriction (150µm × 150µm). Include a lateral cavity (500µm × 80µm) near the constriction to trap a microbubble [1].
- Preparation: Infuse liquid to trap an air pocket in the lateral cavity, forming a quasi-cylindrical microbubble.
- Actuation: Affix a piezotransducer to the microchip and connect to a function generator. Actuate near the bubble's resonant frequency (typically 10-100kHz).
- Testing: Introduce particle suspension at operational flow rates. Test three operational modes:
- Continuous: Constant actuation of microbubble
- Periodic: Intermittent actuation at set intervals
- Event-triggered: Activation only when clogging is detected
- Validation: Compare clogging frequency and duration with and without microstreaming active. Effective implementation should show immediate disruption of developing clogs [1].
Protocol 2: CFD Simulation for Trap Optimization
Purpose: Use Computational Fluid Dynamics to optimize trap geometry before fabrication.
Materials:
- CFD software (ANSYS Fluent, COMSOL Multiphysics, or similar)
- CAD software for geometric design
- Workstation with sufficient processing power
Methodology:
- Model Setup: Create a 3D model of your proposed trap geometry. Include all relevant dimensions - channel width/height, constriction size, and chamber volume.
- Parameter Definition:
- Define fluid properties (density, viscosity) matching your culture medium
- Set boundary conditions (inlet flow rate, outlet pressure)
- For particle tracing, define particle size based on your cells (e.g., 15-17µm for K562 and NIH3T3 cells) [45]
- Mesh Generation: Create a sufficiently fine mesh to resolve flow profiles in constricted regions.
- Simulation Execution:
- Run steady-state simulations to determine flow resistance ratios between trap and bypass paths
- Perform transient particle tracing to visualize cell paths through the device
- Analysis:
- Verify that empty traps have lower flow resistance than bypass channels
- Confirm that once a "cell" (simulated particle) occupies a trap, the resistance ratio reverses
- Ensure particles follow deterministic paths to trapping sites [45]
- Iteration: Modify geometry based on simulation results and re-simulate until optimal performance is predicted.
Research Reagent Solutions
Table 4: Essential Materials for Microfluidic Single-Cell Analysis
| Reagent/Material | Function | Example Application | Key Considerations |
|---|---|---|---|
| Polydimethylsiloxane (PDMS) [44] | Primary chip material; biocompatible, transparent | Device fabrication | Gas permeable; can absorb small molecules |
| Polystyrene Particles [1] | Clogging testing, system validation | Anti-clogging experiments | Available in various sizes; fluorescent labeling enables visualization |
| Sulfate-Stabilized Microspheres [1] | Testing particle flow with minimal adhesion | Clogging studies | Negative charge prevents agglomeration |
| Glycerol Solution [1] | Adjusts medium density for neutral buoyancy | Particle suspension preparation | 23.8% w-w% glycerol matches polystyrene density |
| Gaseous Microbubbles [1] | Core element for active anti-clogging | Microstreaming applications | Air or argon; size controlled by cavity dimensions |
System Workflow and Pathway Diagrams
Microfluidic Experiment Workflow with Integrated Troubleshooting
Deterministic Hydrodynamic Trapping Principle
In microfluidic single-cell analysis research, the integrity of your data is directly dependent on the quality of the initial cell suspension. Clogged channels, inconsistent data, and experimental failure are often traced back to poor sample preparation. This guide provides targeted troubleshooting and best practices to prepare clean, viable single-cell suspensions that prevent microfluidic device blockages and ensure robust, reproducible results.
FAQs: Addressing Common Challenges
1. What are the critical quality metrics for a cell suspension in microfluidics?
A high-quality cell suspension must meet three key standards [47]:
- Clean: The suspension must be free from debris, cell aggregates (clumps), and other contaminants like background RNA or EDTA [47].
- Healthy: Cell viability should be at least 90% to ensure high-quality single-cell data. Lower viability increases debris, which can clog channels and skew results [47] [48].
- Intact: Cellular membranes must be intact. Leaky or lysed cells release RNA and intracellular content, increasing background noise and the risk of microfluidic obstruction [47].
2. My sample has low viability and high debris. How can I clean it up?
Several techniques are available for dead cell and debris removal. The choice depends on your cell type, sensitivity, and available equipment.
- Density Gradient Centrifugation: Separates cells based on size and density. Dead cells and debris are less dense and will form a separate layer. While simple, high centrifugation speeds can damage or lyse fragile cells [49] [48].
- Magnetic-Activated Cell Sorting (MACS): Uses antibody-bound magnetic beads to target and remove dead cells (which often express phosphatidylserine on their surface). It is an effective method but can have lower throughput, and the magnetic field may stress very delicate cells [49] [48].
- Buoyancy-Activated Cell Sorting (BACS): A gentle alternative that uses microbubbles conjugated to Annexin V to bind dead cells and float them to the surface for removal. This method minimizes shear stress, making it ideal for rare or fragile cell populations [49].
- Fluorescence-Activated Cell Sorting (FACS): Can be used to precisely sort and collect live cells based on viability dyes. It is highly accurate but requires specialized, expensive equipment and carries a risk of cell shearing due to high flow pressures [49] [48].
Table 1: Comparison of Debris and Dead Cell Removal Methods
| Method | Principle | Best For | Throughput | Relative Cost | Gentleness |
|---|---|---|---|---|---|
| Density Centrifugation | Size/Density separation | Routine samples, first-step cleanup | Medium | Low | Low |
| Magnetic Sorting (MACS) | Antibody-magnetic bead binding | Specific population depletion | Medium | Medium | Medium |
| Buoyancy Sorting (BACS) | Microbubble flotation | Rare, fragile, or sensitive cells | High | Medium | High |
| Flow Sorting (FACS) | Fluorescence-based detection | High-precision live cell isolation | High | High | Low-Medium |
3. How do I prevent cells from clumping together in suspension?
Cell clumping is a primary cause of microfluidic clogging. To prevent aggregation [48]:
- Use appropriate buffers: Phosphate-buffered saline (PBS) without calcium and magnesium prevents clumping. Adding 0.04%–1% Bovine Serum Albumin (BSA) or 1%–10% Fetal Bovine Serum (FBS) can reduce non-specific binding.
- Add DNase I: DNA released from dead cells can entangle live ones into clumps. Adding DNase I enzyme degrades this free DNA, preventing aggregation [48].
- Use gentle pipetting techniques: Always use wide-bore pipette tips to minimize shear stress that can damage cells and release more DNA [47].
- Keep samples cold: Store prepared cell suspensions on ice to slow down metabolic activity and adhesion, unless the cell type is sensitive to cold [48].
4. My microfluidic channels keep clogging, but my cell suspension seems fine. What else could be wrong?
The problem may lie in the physical properties of your cells relative to your device design [50] [47]:
- Cell Size: Droplet-based microfluidic devices have a strict upper cell size limit (typically < 30 µm). Cells larger than the droplet (30-40 µm), such as neurons, cardiomyocytes, or hepatocytes, will inevitably clog the channels [50] [47].
- Solution: For large cells, consider switching to single-nuclei RNA sequencing (snRNA-seq) or using an instrument-free combinatorial barcoding technology that is not constrained by cell size [50].
Troubleshooting Guides
Problem: Persistent Cell Clustering and Aggregation
Cell clusters are a major cause of blockages in channel constrictions.
Table 2: Troubleshooting Cell Clustering
| Symptom | Possible Cause | Solution |
|---|---|---|
| Visible clumps after resuspension | DNA from dead cells causing stickiness | Add DNase I (0.1-1 U/mL) to your wash buffer to digest extracellular DNA [48]. |
| Cells clump after centrifugation | Over-concentrated pellet; harsh pipetting | Resuspend gently with wide-bore pipette tips. Avoid excessive centrifugation force and time [47]. |
| Clumping in specific media | Buffer composition issues | Use a PBS-based buffer without Ca2+/Mg2+ and supplement with 0.04%-1% BSA to reduce cell adhesion [47] [48]. |
| Tissue-derived cells are fibrous | Incomplete tissue dissociation; myelin or ECM | For brain tissue, add a myelin removal step. Optimize enzymatic digestion (e.g., with hyaluronidase) to break down sticky ECM components [50] [51]. |
Problem: High Debris Content and Low Viability
Excessive debris and dead cells can adhere to channel walls and narrow constrictions, leading to progressive blockage.
Table 3: Troubleshooting Debris and Viability
| Symptom | Possible Cause | Solution |
|---|---|---|
| Low viability after thawing | Cryo-damage during freezing/thawing | Start with >90% viability before freezing. Use cryoprotectants like DMSO and freeze cells slowly. Thaw quickly and wash away cryoprotectant immediately [48]. |
| High debris in tissue samples | Overly harsh mechanical or enzymatic dissociation | Tailor the dissociation protocol to the tissue. Use gentler enzymatic cocktails (e.g., a mix of collagenase and dispase) and limit mechanical force [50] [51]. |
| General low viability | Toxic reagents or prolonged handling | Limit exposure to EDTA and surfactants. Keep processing times short and perform steps on ice where possible to preserve cell health [48]. |
Experimental Protocols for Prevention
Detailed Protocol: Enzymatic Debris Reduction with DNase I
This protocol is effective for reducing clumping caused by genomic DNA release [48].
- Prepare DNase Solution: Dilute DNase I enzyme in your cell suspension buffer (e.g., PBS + 0.04% BSA) to a final working concentration of 0.1-1 U/mL.
- Wash Cells: Centrifuge your cell suspension and gently decant the supernatant.
- Treat Pellet: Gently resuspend the cell pellet in the prepared DNase I solution using a wide-bore pipette tip.
- Incubate: Allow the cells to incubate in the solution for 15-20 minutes at room temperature or 37°C, depending on cell sensitivity.
- Stop Reaction: Add a stop solution (often containing EDTA) or wash the cells twice with a large volume of buffer to remove the DNase and digested DNA fragments.
- Resuspend: Resuspend the final, clean cell pellet in an appropriate running buffer for your microfluidic device.
Advanced Anti-Clogging Microfluidic Technique: 3D Microbubble Streaming
For persistent clogging at specific channel constrictions, active anti-clogging technologies can be integrated. One innovative method uses 3D microbubble streaming [1].
- Principle: A microbubble is trapped in a cavity adjacent to a microchannel constriction. When activated by a piezotransducer at resonant frequency, the bubble oscillates, generating strong, counter-rotating micro-vortices in the surrounding fluid [1].
- Effect: This microstreaming creates high shear stress and a 3D velocity gradient that effectively inhibits the formation of particle arches (clogs) at the constriction and can break apart existing clusters [1].
- Implementation: The system can be operated in event-triggered, continuous, or periodic modes, offering flexibility for different applications. This provides a powerful, non-invasive, and biocompatible real-time solution to clogging [1].
The following diagram illustrates the decision-making workflow for diagnosing and addressing clogging issues in microfluidic systems.
The Scientist's Toolkit: Essential Reagents & Materials
Table 4: Key Research Reagent Solutions for Cell Preparation
| Reagent/Material | Function & Application |
|---|---|
| DNase I | Enzyme that degrades extracellular DNA, effectively reducing cell clumping and aggregation [48]. |
| Wide-Bore Pipette Tips | Minimizes shear stress during pipetting, protecting fragile cells from damage and preventing debris formation [47]. |
| PBS + BSA Buffer | A physiological buffer without Ca2+/Mg2+ to prevent clumping. BSA coats cells and reduces non-specific sticking [47] [48]. |
| Dispase | A neutral protease ideal for gentle detachment of cell colonies and dissociating tissues into small clumps by targeting collagen IV and fibronectin [51]. |
| Collagenase | Enzyme critical for digesting the extracellular matrix in solid tissues, particularly those rich in collagen [51]. |
| Hyaluronidase | Breaks down hyaluronic acid in the ECM, especially useful for tough tissues like brain and tumors [50] [51]. |
| Dead Cell Removal Microbubbles (BACS) | Microbubbles conjugated to Annexin V bind phosphatidylserine on dead cells, enabling their gentle removal via flotation [49]. |
| TrypLE | A gentle enzyme reagent for dissociating adherent cells (e.g., cell lines) without altering surface antigen expression as trypsin can [50] [51]. |
Emergency Response Protocols for Acute Blockage Scenarios
FAQ: Addressing Microfluidic Channel Blockages
What are the immediate signs of a partial or complete channel blockage? A sudden, sustained increase in pressure or a drop in flow rate to zero are primary indicators. Visually, you may observe an accumulation of debris or cells at a specific point in the channel, or a halt in droplet generation in droplet-based systems [12].
What should I do first when a blockage is suspected?
- Stop the flow immediately to prevent further buildup of material.
- Do not exceed the pressure limit of your device, as this can cause catastrophic failure.
- Attempt a gentle reversal of flow direction if your system allows it. This can often dislodge the obstruction without damaging the chip.
How can I prevent blockages in single-cell RNA sequencing workflows? Ensuring a single-cell suspension is critical. Always filter your cell suspension through an appropriate cell strainer before loading it into a microfluidic device. For droplet-based systems like Drop-seq or inDrop, verify that the cell concentration is optimized to minimize doublets and avoid empty droplets, which also reduces the risk of channel clogging [12].
What are the recommended methods for sterilizing microfluidic devices to prevent contamination-related issues? For lab-scale prototyping, ethanol or isopropanol rinsing are common. However, for commercial or clinical applications, you must use an established sterilization method. Radiation sterilization (E-beam or Gamma) is highly effective for thermoplastics, leaves no residue, and penetrates micron-scale channels without issue. Chemical methods like Hydrogen Peroxide Gas Plasma are also suitable, though they may have penetration limitations in very small capillaries [52].
What safety protocols should be followed when performing these procedures? Always wear appropriate Personal Protective Equipment (PPE), including closed-toe shoes. Be aware of the chemicals and biological materials you are using and notify facility directors of any hazardous materials. No food or beverages are allowed in the lab facility [53].
Troubleshooting Guide: Acute Blockage Scenarios
Diagnosis and Resolution Workflow
The following diagram outlines the logical steps for diagnosing and responding to a channel blockage.
Quantitative Data for Blockage Resolution
Table 1: Chemical Flushing Solutions for Blockage Clearance
| Solution | Concentration | Target Blockage Type | Incubation Time | Key Consideration |
|---|---|---|---|---|
| Sodium Hydroxide | 0.1 - 1.0 M | Proteins, Lipids, DNA | 15 - 30 minutes | Corrosive; check polymer compatibility [52]. |
| Hydrogen Peroxide | 1 - 3% | Biological residues, organics | 10 - 20 minutes | Strong oxidizer; may degrade surface chemistry. |
| Bleach (NaClO) | 1 - 10% | Biological residues | 5 - 15 minutes | Highly corrosive; use with extreme caution and short contact times. |
| HCl | 1 - 5% | Salt precipitates, inorganic crystals | 5 - 10 minutes | Highly corrosive; effective on mineral deposits. |
| Tween-20 / SDS | 0.1 - 1% | Hydrophobic aggregates, lipids | 30 - 60 minutes | Gentle surfactant; good for preventative flushes. |
| DNase I / Protease | As per supplier | Nucleic Acids / Proteins | 30 - 60 minutes | Enzymatic; specific for biological polymer clogs. |
Table 2: Comparison of Re-sterilization Methods for Cleared Devices
| Method | Typical Cycle Time | Material Compatibility (Thermoplastics) | Efficacy for Microfluidics | Regulatory Status |
|---|---|---|---|---|
| Ethanol Rinse | 5-10 min | Good | Good for lab use | Novel [52] |
| UV Treatment | 15-30 min | Good | Surface sterilization only | Novel [52] |
| Autoclave (Steam) | 20-60 min | Poor (deformation risk) | High | Established (Category A) [52] |
| Ethylene Oxide (EtO) | 1-6 hours | Good | High (penetration concerns) | Established (Category A) [52] |
| Hydrogen Peroxide Gas Plasma | ~75 min | Good | Medium (capillary penetration) | Established (Category A) [52] |
| E-beam Radiation | Seconds-Minutes | Good to Excellent | High (excellent penetration) | Established (Category A) [52] |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Blockage Prevention and Management
| Item | Function | Application Note |
|---|---|---|
| Cell Strainer | Removes cell clumps and large debris from suspension before loading. | The most critical step for preventing blockages in single-cell protocols [12]. Use 30-40 µm filters. |
| BSA or Serum Albumin | Passivates channel surfaces to reduce non-specific adhesion of cells and biomolecules. | A standard pre-treatment to minimize protein and cell sticking. |
| Tween-20 or Pluronic F-127 | Non-ionic surfactants that reduce surface adhesion and stabilize droplet interfaces. | Essential for droplet-based scRNA-seq (e.g., Drop-seq, inDrop) and as a preventative flush additive [12]. |
| DNase I / RNase A | Enzymes that degrade nucleic acids which can form viscous clogs from lysed cells. | Use in post-experiment cleaning protocols to break down specific biopolymer blockages. |
| SDS (Sodium Dodecyl Sulfate) | Ionic detergent for powerful cleaning of organic and proteinaceous residues. | Effective for clearing severe biological clogs; requires thorough rinsing. |
| EDTA | Chelating agent that binds divalent cations (Ca2+, Mg2+). | Prevents precipitation of salts and can help disrupt biofilm structures. |
| Filtered Buffers & Reagents | Removes particulate matter from all liquids before entering the microfluidic system. | Always pressure-filter (0.2 µm) buffers, oils, and surfactant solutions. |
Integration of Fail-Safe Mechanisms and Redundant Channel Designs
Troubleshooting Guides and FAQs
This technical support resource addresses common challenges in microfluidic single-cell analysis, with a focus on preventing channel blockage to ensure experimental reliability and data integrity.
Frequently Asked Questions (FAQs)
Q1: What are the most common causes of channel blockage in single-cell analysis chips? Channel blockage most frequently occurs due to the presence of cellular aggregates or debris in the sample. In single-cell RNA-sequencing platforms like Hydro-Seq, despite an initial enrichment step, residual contaminating cells (erythrocytes and leukocytes) can obstruct channels. The design incorporates specific cell capture sites (e.g., 10 × 10 µm openings) to trap larger cells like Circulating Tumor Cells (CTCs) while allowing smaller blood cells to pass through, but sample preparation remains critical [54].
Q2: How can I prevent bubble formation, which can block channels and disrupt flow? Bubble formation is a significant safety and operational concern that can cause pressure fluctuations and unpredictable fluid behavior. To mitigate this, ensure all reagents are properly degassed before loading them into the system. Furthermore, operating the system with precise pressure controls and incorporating bubble traps into the design can help prevent formation and remove any bubbles that do occur [55].
Q3: My passive check valves are leaking. What could be the issue? Leakage in passive check valves often indicates that the valve's diodicity (the ratio of forward to reverse flow resistance) is too low. Valves with planar designs, such as flap or plug valves, are engineered for high diodicity and can be less prone to leakage. Verify that the operating pressure is within the valve's specified range and check for any damage to the valve's movable components, such as the flap, membrane, or ball [56].
Q4: What is the benefit of using a redundant channel design? Redundant channel designs enhance system reliability by providing alternative flow paths. If a channel becomes blocked, fluid can be automatically rerouted through a parallel channel. This is often managed by integrated switch valves, which act as dynamic controllers to open, close, or change fluidic pathways, ensuring the experiment continues without interruption [57].
Q5: How do the pneumatic (Quake) valves in my chip function as a fail-safe? Pneumatic valves control flow by using external pressure to deform an elastomeric membrane (often PDMS) to open or close a channel. In fail-safe scenarios, these valves can be designed to default to a "closed" state in the event of a pressure loss, securely isolating sensitive areas of the chip. In the Hydro-Seq platform, these valves are used to selectively close flow paths and isolate capture chambers during critical washing and mRNA extraction steps, preventing cross-contamination [54].
Q6: Why is my device's performance inconsistent, and how is this a safety issue? Inconsistency can stem from manufacturing variations in microscale channel dimensions, surface properties, or bonding quality. These variations lead to unpredictable fluid behavior. From a safety and compliance perspective, such inconsistencies can compromise the validity of diagnostic results, leading to errors. Adherence to stringent quality control protocols and manufacturing tolerances is essential to ensure device reliability and user safety [55].
Quantitative Data on Fail-Safe Mechanisms
The table below summarizes key metrics for components that prevent or mitigate channel blockage.
| Component | Function | Key Performance Metric | Reported Value / Specification |
|---|---|---|---|
| Planar Check Valves [56] | Prevents backflow; maintains unidirectional flow | Diodicity | High (specific ratio depends on design) |
| Cell Capture Site [54] | Hydrodynamically captures single cells; allows debris to pass | Capture Opening Size | 10 × 10 µm |
| On-Chip Washing (Hydro-Seq) [54] | Removes cellular contaminants from chambers | Contaminant Reduction | Two orders of magnitude |
| High-Efficiency Cell Capture [54] | Ensures rare cells are captured for analysis | Single-Cell Capture Efficiency | 72.85% ± 2.64% (mean ± SD) |
| Microvalve with Ultra-Low Carryover [57] | Minimizes cross-contamination between reagents | Carryover Volume | 1.5 µl |
| Zero Dead Volume Microvalves [57] | Eliminates areas where fluid can be trapped and cause clogs | Dead Volume | Zero |
Experimental Protocol: On-Chip Contaminant Washing and Cell Lysis
This protocol is adapted from the Hydro-Seq platform for achieving contamination-free single-cell RNA-sequencing [54].
1. Cell Loading and Initial Capture:
- Objective: To hydrodynamically capture single cells in dedicated chambers.
- Method: Introduce the enriched cell sample into the chip's inlet. Apply a negative pressure at the outlet via a syringe pump to withdraw the sample through the chip. During this phase, the bead flow channels and washing channels are kept closed by pneumatic Quake valves. Cells are captured at the 10 × 10 µm cell capture sites within each chamber. Once a cell occupies a site, it blocks that flow path, diverting subsequent cells to downstream chambers.
2. On-Chip Washing for Contaminant Removal:
- Objective: To remove residual contaminating cells (e.g., leukocytes, erythrocytes) and debris from the capture chambers.
- Method: After initial cell loading, open the bead capture sites. This allows a washing buffer (e.g., Phosphate-Buffered Saline - PBS) to flow through the chambers, carrying away any non-captured contaminants.
- Enhanced Washing (Reloading): To further reduce contaminants in the dead volume, retrieve the captured cells by flushing 100 µL of PBS into the inlet. Repeat the cell loading step with this diluted sample. This process dilutes the contaminant concentration by approximately two orders of magnitude, which is then followed by a second wash.
3. Bead Pairing and Chamber Isolation:
- Objective: To introduce barcoded beads for mRNA capture and isolate individual reaction chambers.
- Method: Load a suspension of barcoded beads (average diameter ~40 µm) into the chip. The beads are captured in bowl-shaped bead capture pockets (20 × 25 µm openings) within each chamber, pairing them with the captured cells. Once paired, close all pneumatic valves connected to the chambers to isolate them from each other and the main flow channels.
4. Cell Lysis and mRNA Hybridization:
- Objective: To lyse the captured cells and hybridize the released mRNA onto the barcoded beads.
- Method: Introduce a cell lysis buffer into the branch channels via the washing channels. The valves isolating the chambers are then briefly opened. Due to the square shape of the bead capture opening and the round bead, a small leakage flow is generated, introducing the lysis buffer into the chambers. The captured cell blocks and seals its site, remaining in place. When the valves are re-closed to isolate the chambers, the action generates a turbulent flow that lyses the cell. The released mRNA molecules hybridize with the capture oligonucleotides on the barcoded beads during a 20-minute incubation period.
5. Bead Retrieval:
- Objective: To collect the beads for downstream sequencing.
- Method: Open all pneumatic valves and apply a backflow to retrieve the barcoded beads from the chambers through the outlet port. The subsequent steps (reverse transcription, amplification, library preparation) follow standard single-cell RNA-sequencing protocols.
Workflow Diagram
The Scientist's Toolkit: Key Research Reagent Solutions
The table below lists essential materials and their functions for the experiments cited.
| Item | Function / Explanation |
|---|---|
| Polydimethylsiloxane (PDMS) [54] | An elastomeric polymer used to fabricate the microfluidic chip via soft lithography. It is gas-permeable and biocompatible, making it suitable for cell culture. |
| Barcoded Beads [54] | Micron-sized beads (e.g., ~40 µm) functionalized with oligonucleotides containing unique molecular identifiers (UMIs). They capture mRNA from a single lysed cell for subsequent single-cell RNA-sequencing. |
| Phosphate-Buffered Saline (PBS) [54] | An isotonic buffer used for on-chip washing procedures to remove cellular contaminants and debris without damaging the captured cells of interest. |
| Lysis Buffer [54] | A chemical solution designed to rupture (lyse) captured cells, thereby releasing intracellular contents, including mRNA, for hybridization with the barcoded beads. |
| Dofetilide [58] | A selective hERG1 potassium channel blocker used in cancer research. Chronic application (e.g., 100 nM for 2-3 weeks) has been shown to reverse a transformed phenotype in hERG1-expressing cells, illustrating its research potential in oncology. |
| PTFE (Polytetrafluoroethylene) [57] | A chemically inert polymer used for valve plugs in commercial microvalves. Its high compatibility with most solvents ensures minimal reaction with reagents and long device life. |
| PEEK (Polyether Ether Ketone) [57] | A high-performance polymer with outstanding mechanical and thermal properties, used for valve seats in microfluidic systems to ensure reliability in demanding applications. |
Performance Benchmarking: Evaluating Anti-Blockage Efficacy Across Platforms
Standardized Metrics for Clogging Resistance and System Reliability
Microfluidic technology has become a cornerstone of modern single-cell analysis, enabling high-resolution insights into cellular heterogeneity that are pivotal for fields like drug development and cancer research [10]. However, the successful implementation of these technologies is frequently compromised by channel clogging and system leaks, which can halt experiments, waste precious samples, and produce unreliable data [59]. This technical support center provides researchers with standardized methodologies and troubleshooting guides to proactively prevent, detect, and resolve these critical issues, thereby ensuring the integrity and reproducibility of your single-cell research.
Frequently Asked Questions (FAQs)
FAQ 1: What are the primary causes of clogging in microfluidic single-cell analysis? Clogging typically occurs due to particulate contamination, cell aggregation, or the formation of air bubbles. In single-cell RNA sequencing workflows, such as Drop-seq and inDrop, the encapsulation of cells into droplets is statistically governed by Poisson distribution, which can lead to undesirable multi-cell encapsulations and potential channel blockages if cell concentration is not optimized [12].
FAQ 2: How can I quickly detect a leak in my microfluidic device? Leaks can occur at component interfaces, within the microchannels, or across the material itself [59]. A simple and effective qualitative method is the visual bubble test: submerge the assembled device in a liquid (e.g., deionized water) and apply a low pressure of air or an inert gas like nitrogen to the fluidic inlet. The formation of a steady stream of bubbles will pinpoint the location of any leak.
FAQ 3: What is the relationship between fluidic resistance and clogging? Fluidic resistance (Rfluidic) is a key parameter in microfluidic design. According to the Hagen-Poiseuille equation, the pressure drop (ΔP) required to maintain a flow rate (Q) is directly proportional to the fluidic resistance: ΔP = Rfluidic * Q [59]. A partial clog dramatically increases the local fluidic resistance of a channel. This means that to maintain the same flow rate, your pressure source (e.g., syringe pump) must work much harder, or the flow rate will drop significantly, disrupting your experiment.
FAQ 4: Are there standardized methods for quantifying clogging and leakage? While universal standards for microfluidics are still emerging, methods from other fields can be adapted [59]. Quantitative metrics for leakage can include:
- Pressure Decay Rate: Measuring the rate of pressure loss in a sealed system.
- Volumetric Leak Rate: Quantifying the volume of fluid lost over time. For clogging, a key quantitative metric is the Normalized Resistance Increase (NRI), which can be calculated from pressure and flow rate measurements.
Standardized Metrics and Measurement Protocols
Table 1: Quantitative Metrics for Clogging and Reliability
| Metric | Formula / Measurement Method | Acceptable Threshold (Example) | Key Application |
|---|---|---|---|
| Fluidic Resistance (Rfluidic) [60] | Rfluidic = ΔP / Q (Measured via meniscus tracking or conductivity) | < 10% deviation from designed value | Baseline performance verification |
| Normalized Resistance Increase (NRI) | NRI = (Rfinal - Rinitial) / R_initial | < 0.5 per hour of operation | Direct quantification of clogging severity |
| Pressure Decay Rate [59] | ΔP/Δt (Measured with a pressure sensor on a sealed, filled device) | < 1% of operating pressure per minute | Seal and material integrity testing |
| Volumetric Leak Rate [59] | ΔV/Δt (Measured by fluid loss in a reservoir or at connections) | Negligible (e.g., < 0.1% of total system volume per hour) | Safety and sample loss risk assessment |
| Single-Cell Encapsulation Efficiency [12] | % of droplets containing exactly one cell (from microscopy or sequencing data) | > 60% (to minimize doublets and empty droplets) | Optimization of cell suspension concentration for droplet-based scRNA-seq |
Protocol 1: Measuring Fluidic Resistance via Conductivity
This protocol provides a highly accurate method for measuring the fluidic resistance of your microchannels, which serves as a critical baseline for detecting clogs [60].
- Solution Preparation: Fill the microchannel with a conductivity standard solution (e.g., 100,000 μS cm⁻¹) [60].
- Electrical Circuit Setup: Create a simple series circuit with a known electrical resistor and the fluid-filled channel. Use a standard voltage meter to measure the voltage drop across the known resistor [60].
- Calculation: Calculate the electrical resistance of the channel (Relec) using voltage divider principles. Then, convert this to fluidic resistance (Rfluidic) using the established relationship: Rfluidic = (8 η L²) / (π A² * Relec * σ), where η is fluid viscosity, L is channel length, A is cross-sectional area, and σ is the solution conductivity [60].
Protocol 2: Pressure Decay Test for Leak Quantification
This protocol standardizes the process for detecting and quantifying leaks [59].
- Setup: Seal all inlets and outlets of the dry microfluidic device. Connect a pressure sensor and a regulated pressure source to one port.
- Pressurization: Fill the device with a test gas (e.g., air, nitrogen) to a predetermined test pressure, typically 1.5x the normal operating pressure.
- Measurement: Isolate the pressurized system and monitor the pressure with the sensor over a set period (e.g., 10-15 minutes).
- Analysis: Calculate the pressure decay rate (ΔP/Δt). A rate exceeding your defined threshold (see Table 1) indicates a leak that requires investigation.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for Microfluidic Reliability
| Item | Function | Application Note |
|---|---|---|
| Polydimethylsiloxane (PDMS) [60] | Common elastomer for soft lithography; gas-permeable, optically clear. | Prone to absorption of small molecules; surface treatment often required to prevent non-specific adhesion. |
| Surfactant (e.g., Krytox 157-FSH) [60] | Stabilizes emulsion droplets and reduces cell and protein adhesion to channel walls. | Critical for preventing coalescence in droplet-based scRNA-seq (e.g., Drop-seq, inDrop) and minimizing clogging [12]. |
| Conductivity Standard Solutions [60] | Pre-mixed solutions with precisely known electrical conductivity. | Essential for accurately calibrating the conductivity-based fluidic resistance measurement method [60]. |
| NIST-Traceable Pressure Gauge | Provides highly accurate and reliable pressure measurements. | Necessary for performing quantitative pressure decay leak tests and for monitoring pressure fluctuations that indicate clogging. |
| Cell Strainer (e.g., 40 μm) | A physical filter to remove large aggregates and debris from cell suspensions prior to loading. | A simple but critical pre-processing step to prevent introductory clogs from overgrown cell cultures or tissue digests. |
Experimental Workflow for Clogging Resistance Assessment
The diagram below outlines a logical workflow for systematically assessing and troubleshooting clogging issues in a microfluidic device, integrating the metrics and protocols detailed above.
The selection of an appropriate microfluidic system is fundamental to the success of single-cell analysis experiments. The table below provides a comparative summary of Droplet, Valve-Based, and Hydrodynamic systems to guide your choice.
Table 1: Comparative Overview of Microfluidic Single-Cell Analysis Systems
| Feature | Droplet Systems | Valve-Based Systems | Hydrodynamic Trapping Systems |
|---|---|---|---|
| Core Principle | Encapsulates cells in picoliter-to-nanoliter droplets within an immiscible continuous phase [61] [62] | Uses pneumatic microvalves (e.g., Quake valves) to compartmentalize cells in microchambers [63] | Employs channel geometry and hydrodynamic forces to physically trap cells at designated sites [27] |
| Throughput | Very High (can exceed 10,000 droplets per second) [61] | Moderate (typically 1,000 - 5,000 chambers per device) [63] | High (easily scalable to thousands of traps) [27] [20] |
| Single-Cell Isolation Efficiency | Stochastic, follows Poisson distribution; can be improved with fluorescence-activated droplet sorting (FADS) [64] | High and deterministic; suitable for rare cells [63] [20] | High and deterministic; "least flow resistance" design achieves ~90-100% trap occupancy [20] |
| Key Advantages | • Extreme parallelization• Minimal reagent consumption• Isolated microreactors prevent cross-contamination [61] [65] | • Precise fluid exchange for multi-step assays (e.g., washing, staining)• On-chip cell culture• Real-time monitoring [63] | • Simple, passive operation (no external fields)• Low cost and easy fabrication• High viability due to low shear stress [27] [20] |
| Common Blockage Challenges | • Nozzle clogging from cell aggregates or debris• Surfactant precipitation [61] | • Valve membrane failure or deformation• Debris accumulation in complex channel networks [63] | • Clogging by larger cell aggregates or debris at trap inlets• Shear-induced damage if design is not optimized [27] |
The following diagram illustrates the core operational workflow and logical relationship for selecting and troubleshooting these systems.
Troubleshooting Guides and FAQs
This section addresses the most frequent issues researchers encounter with each platform, with a focus on preventing and resolving channel blockages.
Droplet System Troubleshooting
FAQ: My droplet generation device keeps clogging. How can I prevent this?
Clogging at the flow-focusing or T-junction nozzle is a common failure point. Preventive measures and solutions are multi-faceted.
- Q: What are the primary causes?
- A: The presence of cell clusters or aggregates larger than the nozzle diameter, or the precipitation of surfactants in the oil phase over time.
- Q: What sample preparation steps are critical?
- A: Always filter your cell suspension using an appropriate cell strainer (e.g., 35-40 µm) immediately before loading it into the device. This removes aggregates that are the primary cause of clogs [64].
- Q: How can I maintain my reagents?
- A: Prepare fresh surfactant-containing oil phases and avoid repeated freeze-thaw cycles. If precipitation is suspected, filter the oil phase through a 0.2 µm syringe filter.
- Q: My device is clogged. What can I do?
- A: Applying a brief, reverse flow pulse can often dislodge a recently formed clog. If the clog persists, the device must be discarded and replaced.
FAQ: My droplet generation has become unstable and non-uniform. Why?
- Q: What does unstable generation indicate?
- A: This typically points to an imbalance in the flow rates of the dispersed (aqueous) and continuous (oil) phases, or a partial clog beginning to form.
- Q: What is the first check?
- A: Verify the stability of your pressure or syringe pump system. Ensure there are no air bubbles in the tubing or chip inlets, as they compress and cause flow fluctuations.
- Q: How should I adjust the flow rates?
- A: Refer to your device's design parameters. Generally, a higher continuous-to-dispersed phase flow rate ratio promotes stable droplet generation. Systematically adjust the rates while monitoring droplet formation.
Valve-Based System Troubleshooting
Sealing failure prevents single-cell compartmentalization and is often related to valve integrity or pressure.
- Q: What are the main reasons for leakage?
- A: The actuation pressure may be too low, the PDMS membrane may be ruptured, or the bonding between device layers may have failed.
- Q: How do I optimize the sealing pressure?
- A: The required pressure is highly dependent on valve geometry. Circular valves with a larger radius (e.g., 140 µm vs. 120 µm) require lower sealing pressure (e.g., 0.07 MPa vs. 0.13 MPa). Consult device design specifications [63].
- Q: How can I prevent valve damage?
- A: Avoid applying pressures far above the critical "rupture pressure" determined during design modeling. Use hydraulic actuation with liquid if air permeability through PDMS is an issue [63].
FAQ: The flow channels in my valve device are clogging, especially near the traps.
- Q: Why do these clogs occur?
- A: Debris can accumulate in the complex network of channels, and cell clumps can obstruct narrow constrictions leading to trap sites.
- Q: What is the prevention protocol?
- A: As with droplet systems, rigorous sample filtration is essential. Furthermore, introducing a brief "flush" step with a particle-free buffer through the entire network before engaging the valves can clear initial debris.
- Q: What design feature can mitigate clogging?
- A: Devices designed with a compact layout of concatenated T-junctions reduce the overall channel length and complexity, minimizing areas where debris can settle [20].
Hydrodynamic Trapping System Troubleshooting
FAQ: Cells are bypassing the trap sites and not loading. What should I check?
This issue is directly linked to the fundamental "least flow resistance path" principle.
- Q: Why would a cell bypass a trap?
- A: The flow resistance of the main bypass channel is lower than the resistance of the trap channel. The cell follows the path of least resistance.
- Q: How is this solved in design?
- A: The geometry must be optimized so that an empty trap has a lower flow resistance than the main bypass. When a cell enters the trap, it dramatically increases the trap's resistance, redirecting subsequent cells to the next empty trap [20]. Confirm your device's design was optimized using Computational Fluid Dynamics (CFD) simulation.
- Q: What operational factor could cause this?
- A: An excessively high flow rate can create shear forces that prevent cells from settling into the traps. Gradually reduce the flow rate during the loading phase.
FAQ: My hydrodynamic trap array is clogged with an aggregate. How can I clear it?
- Q: Can I clear a clog without damaging the device?
- A: Often, yes. Applying a controlled reverse flow pulse can flush the aggregate backward and out of the trap inlet.
- Q: How can trap design reduce clogging?
- A: Designs that feature smooth, arc-contoured transitions into the trap, rather than sharp corners, facilitate smoother cell entry and reduce the chance of snagging and clogging [20]. Ensuring the trap inlet is appropriately sized (e.g., 20-25% smaller than the target cell diameter) also helps [27].
Detailed Experimental Protocols
Protocol: High-Efficiency Single-Cell Trapping in a Hydrodynamic Device
This protocol is adapted from a device that achieved 90-100% single-cell trapping efficiency over 400 trap sites [20].
1. Device Priming:
- Flush the entire microfluidic device with a biocompatible buffer (e.g., PBS) to remove any air bubbles and manufacturing debris. Ensure all channels are wetted.
- Treat the device with a surface passivation agent (e.g., 1% Pluronic F-127 in PBS) for at least 30 minutes to prevent non-specific cell adhesion. Flush with cell culture medium before use.
2. Cell Sample Preparation:
- Harvest and centrifuge your cell line (e.g., HeLa or HEK-293T).
- Critical Step: Resuspend the cell pellet and pass the suspension through a sterile 35-40 µm cell strainer to break up and remove aggregates.
- Adjust the cell concentration to approximately 1-5 x 10^6 cells/mL in the appropriate culture medium.
3. Cell Loading and Trapping:
- Load the filtered cell suspension into the device inlet using a syringe pump.
- Initiate Trapping: Begin with a moderate flow rate (e.g., 2-4 µL/min) to fill the traps. The "deterministic" design will sequentially fill trap sites.
- Optimize and Monitor: Observe trapping under a microscope. Once the majority of traps are occupied, reduce the flow rate to a maintenance level (e.g., 0.5-1 µL/min) for long-term culture or imaging. The entire trapping process for 400 sites can be completed in under 10 minutes [20].
Protocol: Implementing Fluorescence-Acted Droplet Sorting (FADS)
This protocol enhances droplet systems by enriching for droplets containing single, viable cells, dramatically reducing noise and cost [64].
1. Cell Staining and Encapsulation:
- Stain your cell population with a viability dye (e.g., Calcein-AM for live cells) or a DNA stain (e.g., Vybrant Green for nuclei).
- Co-encapsulate the stained cells with barcoded microgels and lysis reagent in a flow-focusing droplet generator.
2. Droplet Sorting:
- As droplets flow past a laser detection point, the fluorescence is measured.
- A field-programmable gate array (FPGA) applies a predetermined threshold (e.g., fluorescence signal significantly above background from empty droplets).
- When a target droplet is detected, the FPGA triggers an electrical pulse to electrodes at a sorting junction.
- Via dielectrophoresis, the target droplet is deflected into a "positive" collection channel, while empty or debris-containing droplets are wasted.
3. Picoinjection for Multi-Step Assays:
- To increase detection sensitivity, sorted droplets can enter a picoinjector.
- Here, an electric field disrupts the droplet interface, allowing for the injection of additional reagents (e.g., an optimized reverse transcription mix for scRNA-seq).
- This multi-step processing can increase gene detection rates five-fold compared to single-step methods [64].
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for Microfluidic Single-Cell Experiments
| Item | Function | Application Notes |
|---|---|---|
| PDMS (Polydimethylsiloxane) | The most common material for soft lithography microfabrication. Biocompatible, gas-permeable, and optically clear [63] [66]. | Used for Valve-Based and many Hydrodynamic devices. The base-to-curing agent ratio can be adjusted to modify stiffness [63]. |
| Fluorinated Surfactants | Stabilizes water-in-oil emulsions, preventing droplet coalescence in Droplet Microfluidics [61]. | Essential for droplet stability. Available commercially (e.g., from Ran Biotechnologies). Incompatibility with some organic solvents can cause precipitation. |
| Pluronic F-127 | A non-ionic surfactant used to passivate microchannel surfaces. | Prevents cells and biomolecules from sticking to PDMS walls, crucial for maintaining viability and reducing clogging in all system types [20]. |
| Calcein-AM | Cell-permeant fluorescent dye used as a viability marker. | Converted to green-fluorescent Calcein by intracellular esterases in live cells. Used for Fluorescence-Acted Droplet Sorting (FADS) [64]. |
| Barcoded Microgels / Beads | Solid supports containing unique molecular barcodes for tagging cellular contents (e.g., mRNA). | The core of many high-throughput scRNA-seq methods (e.g., inDrop, 10x Chromium). Allows pooling of many cells for processing while retaining single-cell identity [64]. |
| Collagenase/Dispase | Enzyme blend for tissue dissociation into single-cell suspensions. | Critical upstream sample preparation step. Concentration and incubation time must be optimized for each tissue type to maximize viability and minimize clogs [67]. |
Long-Term Stability Assessment in Continuous Operation Scenarios
Technical Support Center
Frequently Asked Questions (FAQs) and Troubleshooting Guides
Q1: What are the primary causes of microchannel clogging during long-term, continuous single-cell analysis experiments?
Clogging in microchannels primarily occurs due to two mechanisms: the formation of stable, static particle arches at channel constrictions and particle aggregation or adhesion to channel walls [1]. In continuous systems, these events are often accelerated by:
- Particle-to-Constriction Size Ratio: Constrictions with a neck-to-particle size ratio (W/d) that is too small are highly prone to arch formation [1].
- Particle Adhesion: Electrostatic or physical interactions between particles (e.g., biological cells) and the channel material (e.g., PDMS) can lead to aggregation and wall adhesion, gradually narrowing the flow path [1].
- Sedimentation: In passive systems, particles can settle out of suspension due to gravity, accumulating and causing blockages [1].
Q2: What active anti-clogging techniques can be implemented without damaging sensitive biological cells?
Microbubble Streaming is an innovative, biocompatible method proven to prevent and remediate clogs in real-time [1]. This technique involves:
- Mechanism: A microbubble trapped in a side cavity adjacent to a channel constriction is acoustically activated by a piezotransducer. This oscillation generates strong, three-dimensional counter-rotating vortices (microstreaming) that create high shear stresses [1].
- Effect: This streaming flow efficiently inhibits arch formation at constrictions and disintegrates existing particle clusters, preventing blockages [1].
- Biocompatibility: The method is non-invasive and known for its inherent biocompatibility, making it suitable for handling delicate biological cells [1].
Q3: How can I configure the anti-clogging system to best suit my experiment's flow conditions and duration?
The microbubble streaming system offers versatile control strategies to balance effective anti-clogging with minimal interference to your process [1]. You can select from three operational modes:
Table: Operational Modes for Microbubble Streaming Anti-Clogging Systems
| Operational Mode | Description | Best For |
|---|---|---|
| Event-Triggered | The system is activated only when a clog is detected. | Experiments where minimal flow disturbance is critical and clogs are infrequent. |
| Continuous | The anti-clogging mechanism operates without interruption. | High-throughput, critical processes where any clogging incident is unacceptable. |
| Periodic | The system activates at predefined time intervals. | Long-term experiments where continuous operation is unnecessary, helping to conserve energy and reduce potential long-term effects of streaming on cells. |
Q4: What specific experimental protocols should I follow to implement and validate the microbubble streaming anti-clogging technique?
Protocol: Implementing Microbubble Streaming for Clog Prevention
- Microchip Fabrication: Fabricate a straight PDMS microchannel (e.g., 20 mm long) using standard soft lithography techniques. The channel should feature a rectangular cross-section (e.g., 150 µm x 450 µm) that linearly narrows into a constriction (e.g., 150 µm x 150 µm). A dedicated cavity (e.g., 500 µm long x 80 µm wide) must be incorporated adjacent to the constriction to trap the microbubble [1].
- Sample Preparation: Prepare a suspension of your particles or cells. To minimize aggregation and adhesion, use particles stabilized with charged groups (e.g., sulfate groups for negative charge). For neutral buoyancy, suspend particles in a density-matched aqueous solution (e.g., with glycerol) to prevent sedimentation [1].
- System Setup & Actuation: Mount a piezotransducer onto the microchip. Upon liquid infusion, a gas microbubble (e.g., air or argon) will be trapped in the side cavity. Actuate the piezotransducer, typically near the bubble's resonant frequency, to induce microstreaming. The optimal frequency and amplitude should be determined experimentally for your specific setup [1].
- Validation and Monitoring: Use microscopy to visually confirm the formation of counter-rotating vortices near the constriction. Conduct long-term experiments with and without microstreaming activation and compare the incidence of clogging events and total operational runtime before failure to statistically validate the method's effectiveness [1].
Q5: Beyond active methods, what passive design strategies can help mitigate clogging risk?
Passive methods rely on channel geometry to induce specific flow patterns that keep particles suspended:
- Curved Microchannels: Spiral or curved channels generate secondary "Dean flow" vortices, which can exert a lift force on particles, moving them away from walls and preventing adhesion [1].
- Pulsatile Flow: Implementing pulsatile (rather than steady) flow has been shown to significantly delay the onset of microchannel clogging compared to constant flow rates [1].
The Scientist's Toolkit: Key Research Reagent Solutions
Table: Essential Materials for Microfluidic Single-Cell Analysis and Clog Prevention Experiments
| Item | Function / Explanation |
|---|---|
| PDMS (Polydimethylsiloxane) | The most common material for rapid prototyping of microfluidic chips via soft lithography; it is transparent, gas-permeable, and biocompatible. |
| Fluorescent Polystyrene Particles | Often used as cell analogs or for tracking flows; available in various sizes (e.g., 50 µm, 100 µm) and densities. |
| Glycerol Solution | Used to prepare density-matched carrier fluids to achieve neutral buoyancy for particles/cells, drastically reducing sedimentation-driven clogging [1]. |
| Piezoelectric Transducer (Piezotransducer) | The actuation component for active anti-clogging techniques like microbubble streaming; it converts electrical signals into mechanical vibrations [1]. |
| Surface-Charged Particles | Particles stabilized with charged groups (e.g., sulfate) provide electrostatic repulsion between particles and against channel walls, preventing agglomeration and adhesion [1]. |
Experimental Workflow and System Diagrams
The following diagram illustrates the logical workflow for assessing and ensuring long-term stability in a microfluidic system, integrating both preventative measures and reactive troubleshooting.
Assessment and Maintenance of Microfluidic System Stability
This diagram details the core mechanism of the microbubble-based anti-clogging technique, showing the key components and the fluid dynamics involved.
Microbubble Streaming Clog Prevention Mechanism
In microfluidic single-cell analysis, maintaining high throughput and ensuring cell viability are paramount for obtaining biologically relevant data. Channel clogging presents a major challenge to these goals, directly impacting experimental throughput by disrupting flow and potentially compromising cellular integrity through increased shear stress or mechanical damage. This technical support center provides targeted troubleshooting guides and FAQs to help researchers identify, address, and prevent these critical issues in their experiments.
Troubleshooting Guides
Problem: Frequent Channel Clogging During Single-Cell Experiments
| Possible Cause | Diagnostic Check | Recommended Solution | Underlying Principle |
|---|---|---|---|
| Cell Clustering & Aggregation | Visual inspection under microscope for cell clumps at channel inlets or constrictions. | - Pre-filter cell suspension using appropriate cell strainers (e.g., 30-40µm).- Incorporate biocompatible surfactants (e.g., 0.1-1% Pluronic F-68) in carrier fluid to reduce adhesion [1]. | Reduces arch formation at constrictions by ensuring single-cell entry and minimizing cell-cell and cell-wall adhesion. |
| Incorrect Cell Concentration | Calculate expected encapsulation rate/droplet occupancy using Poisson distribution. | Adjust cell concentration to achieve a target occupancy of 10-20% for single cells in droplet-based systems [68]. | Prevents over-crowding and multi-cell encapsulations, which are common precursors to clogs. |
| Channel Geometry & Size | Check if channel/constriction width is appropriate for target cell size (typically 1.5-3x cell diameter). | Redesign chip with constrictions sized appropriately for your cells (e.g., W/d ratio of 3 for 50µm particles) [1]. | Prevents physical jamming of cells or large clusters at narrow channel segments. |
| Particulate Contaminants | Inspect buffers and reagents for visible particles. | Centrifuge and filter all reagents (0.22µm filter) before loading into the system. | Removes external debris that can nucleate clog formation. |
Problem: Reduced Cell Viability Post-Processing
| Possible Cause | Diagnostic Check | Recommended Solution | Underlying Principle |
|---|---|---|---|
| High Shear Stress in Constrictions | Observe cell deformation or lysis in high-speed regions. | - Reduce operational flow rates and pressure.- Use wider channels or gradual constrictions to minimize shear peaks [69]. | Excessive shear forces can damage cell membranes and compromise viability. |
| Prolonged Exposure to Unfriendly Materials | Review material compatibility (e.g., PDMS absorbency). | - Use biocompatible coatings (e.g., PEG) on channel walls.- Consider alternative chip materials (e.g., glass) for sensitive cells [10]. | Minimizes chemical stress and non-specific binding that can impair cell health. |
| Inadequate On-Chip Culture Conditions | Monitor cell morphology over time in trapping or culture regions. | For long-term assays, ensure integrated perfusion of fresh medium and gas exchange (e.g., CO₂) [10]. | Maintains a physiological microenvironment to support cell survival during extended analysis. |
Problem: Sudden Drop in Throughput or Flow Rate
| Possible Cause | Diagnostic Check | Recommended Solution | Underlying Principle |
|---|---|---|---|
| Partial or Complete Clog | Check for pressure spikes or flow cessation in specific sections. | Implement active anti-clogging strategies:1. Microbubble Streaming: Integrate a microbubble cavity near constrictions; activate with piezotransducer to generate scouring vortices [1].2. Flow Pulsation: Switch from steady to optimized pulsatile flow to delay clogging [1]. | Microstreaming generates high shear stress that disrupts particle arches and clusters without damaging cells. |
| Bubble Formation in Channels | Visual identification of air bubbles obstructing flow. | - Degas buffers before use.- Include bubble traps in chip design.- Flush system with degassed buffer or ethanol to dissolve bubbles. | Air bubbles act as physical barriers to fluid flow and cell passage. |
Detailed Experimental Protocols
Protocol 1: Microbubble Streaming for Real-Time Clog Prevention
This protocol utilizes acoustic-activated microbubbles to generate disruptive microvortices, preventing clog formation in a biocompatible manner [1].
Materials:
- PDMS or glass microfluidic chip with integrated lateral cavity (e.g., width: 80µm, length: 500µm) adjacent to the constriction.
- Piezoelectric transducer (Piezotransducer).
- Function generator and amplifier.
- Cell suspension prepared in appropriate buffer.
Method:
- Chip Priming: Flush the microchannel with the carrier fluid. A gas (e.g., air) pocket will be trapped in the lateral cavity, forming a quasi-cylindrical microbubble.
- Actuator Setup: Affix the piezotransducer to the microchip surface near the bubble cavity.
- Frequency Calibration: Use the function generator to apply an acoustic signal. Sweep through frequencies (e.g., 1-20 kHz) while observing the bubble under a microscope to identify its resonant frequency, indicated by maximal oscillatory amplitude.
- Operation Mode Selection: Choose an operational mode based on your application:
- Event-Triggered Mode: Activate the transducer automatically upon detection of a flow rate drop or increased pressure.
- Periodic Mode: Actuate the transducer at set intervals (e.g., 5-10 seconds every minute).
- Continuous Mode: For high-risk scenarios, run the transducer continuously at a calibrated amplitude.
- Experiment Execution: Introduce the cell suspension into the chip at the desired flow rate while the anti-clogging system is active.
Validation: Compare the total particle throughput and duration of uninterrupted operation with and without microbubble activation. Statistical analysis should show a significant delay in clogging incidents [1].
Protocol 2: Optimized Cell Preparation for High-Viability Encapsulation
Proper cell sample preparation is critical to prevent clogging at the source and maintain high viability.
Materials:
- High-viability cell suspension (e.g., >95% viability confirmed by Trypan Blue exclusion).
- Cell strainers (30µm or 40µm).
- Biocompatible surfactant (e.g., Pluronic F-68).
- Centrifuge.
Method:
- Harvesting: Gently dissociate cells to obtain a single-cell suspension. Avoid aggressive pipetting or trypsinization that can induce stress.
- Washing: Centrifuge cells and resuspend in a buffer containing a biocompatible surfactant like 0.1% Pluronic F-68.
- Filtration: Pass the cell suspension through a pre-wetted cell strainer (30-40µm) to remove pre-existing aggregates and debris.
- Concentration Adjustment: Count cells and accurately dilute the suspension to the target concentration for your microfluidic platform (typically 10^5 - 10^6 cells/mL for droplet generators, aiming for ~10% occupancy) [68].
- Loading: Keep the prepared sample on ice or at 4°C until ready to load into the chip to minimize metabolic activity and aggregation.
Anti-Clogging Method Comparison Table
The following table summarizes key characteristics of different clog mitigation strategies.
| Method | Principle | Throughput Impact | Viability Compatibility | Key Advantage |
|---|---|---|---|---|
| Microbubble Streaming [1] | Acoustic actuation creates 3D scouring vortices. | High (Real-time, continuous) | High (Biocompatible) | Non-invasive; can be event-triggered. |
| Pulsatile Flow [1] | Alternating pressure profiles disrupt arch formation. | Medium | High | Can be implemented with modified pump systems. |
| Passive Dean Flow [1] | Curved channels generate helical secondary flows. | Medium | High | No external power required post-fabrication. |
| Hydrodynamic Trapping [69] [68] | Physical structures capture single cells. | Lower (Fixed number of traps) | Medium (Risk of shear at trap) | Simple, well-established passive method. |
| Droplet Isolation [69] [68] | Cells individually encapsulated in oil. | Very High | High | Compartmentalization eliminates cross-contamination. |
Frequently Asked Questions (FAQs)
Q1: My chip is already clogged. Is there a simple way to clear it without damaging the device? Yes, a common method for clearing mild clogs, particularly those caused by cell clusters or hydrophobic polymers, involves flushing with a solvent. Procedure: Using a hand-held syringe, apply as much pressure as possible to flush filtered distilled water or ethanol through the chip. For persistent clogs, after removing any metal components (e.g., needles), place the chip in a standard kitchen microwave oven for ~5 minutes at 500-700 watts. The heating can help break down organic blockages. Reinstall the ports and flush with solvent again [3]. Caution: This method may not be suitable for all chip materials or complex blockages.
Q2: How can I design my microfluidic channels to be less prone to clogging from the start? Consider these design principles:
- Constriction Ratio: Ensure the channel constriction is sufficiently wider than your cell diameter (a W/d ratio of 3 is more robust than 1.5) [1].
- Gradual Tapering: Use linear or curved nozzles with shallow angles (e.g., 45°) leading into constrictions rather than abrupt changes in width.
- Smooth Surfaces: Ensure channel walls are as smooth as possible to prevent cells from snagging.
- Integrated Anti-Clogging Features: Design cavities for microbubbles near critical constrictions to allow for active cleaning [1].
Q3: What are the key quality control indicators I should monitor during a single-cell sequencing run? The core QC indicators are:
- Throughput Maintenance: A stable, high rate of single-cell encapsulation or trapping over time. A sudden drop indicates a developing clog.
- Viability Preservation: Post-processing viability should remain high (e.g., >80-90%, depending on the assay), often confirmed by off-chip staining if not directly measurable on-chip.
- Single-Cell Yield: The percentage of droplets or traps containing exactly one cell. This is optimized by tuning cell concentration according to Poisson statistics [68].
- Data Quality: In sequencing applications, high gene detection counts per cell and low ambient RNA background are indirect indicators of healthy, un-compromised cells.
Q4: Beyond preventing clogs, how can active microfluidics improve my single-cell analysis? Emerging active microfluidic devices use external fields (electrical, optical, magnetic, acoustic) to manipulate single cells with high precision. This allows for:
- Addressable Single-Cell Manipulation: Precisely moving, trapping, and pairing individual cells [10].
- Controlled Microenvironment: Precisely controlling the culture conditions for single cells over time for dynamic studies [10].
- Integrated Analysis: Performing on-chip lysis, nucleic acid amplification, and detection in a highly automated and efficient manner [10].
Signaling Pathways and Workflows
Microbubble Anti-Clogging Mechanism
The following diagram illustrates the mechanism by which acoustically-activated microbubbles prevent clogging in microfluidic channels.
Experimental Workflow for Clog-Resilient Single-Cell Analysis
This workflow integrates preventative measures and quality control checks for a robust single-cell analysis experiment.
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function in Experiment | Key Consideration |
|---|---|---|
| Pluronic F-68 | Non-ionic surfactant added to carrier fluid to reduce cell adhesion and aggregation in channels [1]. | Biocompatible; helps maintain cell viability by minimizing shear stress and non-specific binding. |
| Polystyrene Microspheres | Fluorescent particles of defined size (e.g., 50µm, 100µm) used for calibrating chip performance and testing anti-clogging methods [1]. | Stabilized with charged groups (e.g., sulfate) to prevent agglomeration and mimic cell behavior. |
| Piezoelectric Transducer | External actuator that generates acoustic waves to oscillate microbubbles for microstreaming-based anti-clogging [1]. | Must be compatible with chip material and frequency-tunable to find the bubble's resonance. |
| Cell Strainers (30-40µm) | Pre-filters used during sample preparation to remove large cell clumps and debris before loading the cell suspension into the chip. | Critical pre-processing step to prevent introduction of clogging agents. |
| Agarose Hydrogel | Material used to create hydrogel microspheres for encapsulating single cells in methods like SiC-seq for whole-genome analysis [68]. | Allows diffusion of enzymes and reagents while retaining large DNA fragments. |
| Barcoded Microparticles (Beads) | Functionalized beads with oligonucleotide barcodes used in droplet-based scRNA-seq (e.g., Drop-seq) to label cellular mRNA, enabling multiplexing [68]. | Each bead contains a unique barcode to assign transcripts to their cell of origin. |
Troubleshooting Guide: Microfluidic Channel Blockage
FAQ: What are the primary causes of channel blockage in microfluidic single-cell analysis?
Channel blockage typically occurs due to three main factors: the presence of cell clumps or debris in the sample, the use of cells or beads that are too large for the specific channel dimensions, or the accumulation of proteins and other materials on the channel walls over time.
| Primary Cause | Preventive Measure | Corrective Action |
|---|---|---|
| Cell Clumps/Debris [10] | Implement stringent sample preparation and filtration protocols. | Reverse flow if possible; flush system with appropriate buffer or enzymatic cleaner [10]. |
| Oversized Cells/Beads [12] | Characterize cell size distribution; ensure device geometry is appropriate. | Halt operation; manually clear the inlet reservoir or specific channel segment [10]. |
| Protein/Biofouling [10] | Use surface coatings (e.g., BSA, Pluronic F-68); incorporate regular cleaning cycles. | Flush with a 1% (w/v) SDS solution, 70% (v/v) ethanol, or a 0.5 M NaOH solution [70] [71]. |
FAQ: How can I prevent non-specific adhesion of cells or beads to microchannel walls?
Non-specific adhesion is a common cause of channel blockage and can be mitigated by surface passivation. The following table details effective blocking reagents and their applications [70] [71].
| Reagent Solution | Function / Explanation | Typical Working Concentration |
|---|---|---|
| Bovine Serum Albumin (BSA) | Blocks hydrophobic binding sites on channel surfaces, reducing protein and cell adhesion. | 1% (w/v) |
| Pluronic F-68 | Non-ionic surfactant that adsorbs to surfaces, creating a hydrophilic, protein-resistant layer. | 0.1% - 1% (w/v) |
| Normal Serum | Provides a complex protein mixture to saturate non-specific binding sites; should be from a species different than your detection antibodies. | 2% - 10% (v/v) |
| Polyethylene Glycol (PEG) | Forms a dense, neutral polymer brush layer on surfaces, sterically hindering adhesion. | 0.01% - 1% (w/v) |
FAQ: My droplet-based single-cell encapsulation efficiency is low, leading to wasted reagents. How can I optimize this?
Low encapsulation efficiency in platforms like Drop-seq or 10X Chromium often stems from issues with cell concentration and sample quality [12].
| Problem Source | Test or Action |
|---|---|
| Incorrect Cell Concentration | Accurately count cells and adjust concentration to achieve optimal Poisson distribution for single-cell encapsulation (e.g., ~100,000 cells/mL for many droplet systems) [12]. |
| Cell Clumping | Filter the cell suspension through an appropriate cell strainer (e.g., 30-40 µm) immediately before loading into the microfluidic device [10]. |
| Channel or Nozzle Fouling | Perform a pre-run with a passivation agent (e.g., 1% BSA) and ensure all buffers are particle-free by filtration (0.2 µm) [71]. |
Experimental Protocols for Blockage Prevention
Detailed Methodology: Pre-experiment Surface Passivation for Polydimethylsiloxane (PDMS) Chips
This protocol is designed to minimize protein adsorption and cell adhesion in PDMS-based microfluidic devices, a common source of channel blockage.
- Chip Priming: After standard fabrication and sterilization, fill the microchannels with a 1% (w/v) solution of BSA in 1X PBS.
- Incubation: Allow the BSA solution to incubate within the channels for a minimum of 1 hour at room temperature or 4°C overnight for best results.
- Rinsing: Gently flush the channels with 3-5 channel volumes of 1X PBS or your specific assay buffer to remove unbound BSA immediately before introducing the cell sample.
- Alternative for High-Fouling Samples: For samples prone to severe fouling (e.g., raw biological fluids), consider using a dual-coating strategy. First, incubate with 0.1% (w/v) Pluronic F-68 for 30 minutes, then rinse and follow with the standard BSA passivation protocol [70] [71].
Detailed Methodology: Hydro-Seq for Circulating Tumor Cell (CTC) Analysis from Blood
This valve-based microfluidic method is specifically designed to handle complex samples like whole blood, which are prone to clogging, and to perform high-fidelity scRNA-seq on rare cells [12].
- Sample Preparation: Collect blood in anti-coagulant tubes. Perform red blood cell (RBC) lysis using a commercial lysis buffer. Resuspend the resulting pellet in a buffer containing 0.1% (w/v) BSA to prevent clumping.
- Chip Priming and Loading: Prime the Hydro-Seq chip (a microfluidic device with an array of cell-trapping chambers and integrated valves) with a BSA-containing buffer. Introduce the cell suspension at a controlled, low flow rate to gently load individual cells into the trapping chambers.
- Washing and Lysis: After cell capture, close the microvalves to isolate each chamber. Initiate a flow of lysis buffer across the chip to simultaneously lyse all captured cells and wash away hemoglobin and other PCR inhibitors from the blood sample. This step is critical for preventing biochemical blockage in downstream molecular applications.
- On-chip Barcoding and Library Prep: Perform reverse transcription inside the closed chambers using barcoded primers. Subsequently, pool the barcoded cDNA, amplify it via PCR, and prepare the library for standard next-generation sequencing [12].
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function / Explanation |
|---|---|
| Bovine Serum Albumin (BSA) | A universal blocking agent used to passivate microfluidic channels and prevent non-specific adsorption of proteins and cells [70] [71]. |
| Single-Cell Barcoded Beads | Microbeads (e.g., from Drop-seq or 10X Chromium) functionalized with oligonucleotides containing unique molecular identifiers (UMIs) and cell barcodes to tag mRNA from individual cells [12]. |
| Pluronic F-68 | A non-ionic surfactant used in cell culture and microfluidics to reduce shear stress and prevent surface fouling [71]. |
| Nuclease-Free Water | Essential for preparing all reagent solutions in single-cell RNA-seq workflows to prevent degradation of RNA and DNA oligonucleotides [12]. |
Workflow Diagrams
Microfluidic Single-Cell Analysis Workflow
Channel Blockage Troubleshooting Logic
Cost-Benefit Analysis of Advanced Anti-Clogging Technologies
Clogging presents a major challenge in microfluidic single-cell analysis, leading to inconsistent results, sample loss, and costly operational downtime. This technical support center provides a comprehensive analysis of advanced anti-clogging technologies, offering practical guidance for researchers and scientists engaged in single-cell genomics, drug development, and clinical diagnostics. The following sections present detailed troubleshooting guides, comparative data, and experimental protocols to help you select and implement the most appropriate clogging mitigation strategies for your specific applications.
FAQs: Understanding Anti-Clogging Technologies
1. What are the primary causes of clogging in microfluidic single-cell analysis devices?
Clogging in microfluidic channels primarily occurs due to two mechanisms. First, initial cell adhesion to the microchannel wall surface is driven by long-range adhesive forces, including van der Waals forces and electrostatic double-layer interactions [72]. Second, once initial attachment occurs, cell-to-cell adhesion and aggregation dramatically accelerate, leading to complete channel blockage [72]. This problem is particularly pronounced in devices with narrow constrictions and branched microchannels where fluid velocity increases and cells are accelerated toward channel walls [72].
2. How do passive anti-clogging designs compare to active methods in terms of cost and effectiveness?
Passive anti-clogging designs, such as specialized micropost architectures, prevent clogging through channel geometry without requiring external power or control systems. These designs typically offer lower operational costs and simpler implementation but may provide less adaptability to varying sample conditions. Active methods, such as dielectrophoresis or microbubble streaming, use external energy to disrupt clog formation and can be tuned in real-time but require more complex instrumentation and higher operational costs [72] [1]. The choice between these approaches depends on application requirements, with passive methods often suiting high-throughput, standardized applications and active methods benefiting from research environments with diverse sample types.
3. Can I retrofit existing microfluidic equipment with anti-clogging technologies?
Retrofitting possibilities depend on the specific anti-clogging technology and your existing equipment design. Some methods, like certain acoustic or dielectrophoretic systems, may require substantial instrument modification or specialized components that are not easily integrated into existing platforms [72] [1]. However, design-based solutions incorporating optimized channel geometries or surface treatments may be more readily adaptable to current systems. For commercial instruments, consult manufacturer specifications regarding compatibility with anti-clogging upgrades [73].
4. What are the key performance metrics for evaluating anti-clogging technologies?
Key performance indicators for anti-clogging technologies include device lifespan extension, purity efficiency, sample yield, capture efficiency, and operational flow rate maintenance. For example, one study demonstrated that an anti-clogging method extended device operation from 4 hours to 12 hours without performance deterioration [72]. Additional considerations include cell viability preservation, complexity of operation, and total cost of ownership, including both initial investment and ongoing operational expenses.
Troubleshooting Guides
Problem: Frequent Clogging in Cell Separation Applications
Symptoms: Increased back pressure, reduced flow rate, inconsistent sample processing, complete flow cessation.
Solutions:
- Implement Bypass Channels: Integrate alternative carry-forward paths that allow particles to bypass congested areas. Research shows that designs with dedicated bypass zones can maintain 96% cancer cell capture efficiency at 1 mL/min flow rates while preventing clogging [74].
- Apply Dielectrophoresis (DEP): Integrate interdigitated electrode pairs at branch channel junctions to generate repulsive forces that prevent initial cell attachment. At 20V application, this method has demonstrated 97.23% purity efficiency and 38.95% plasma yield while extending device operation from 4 to 12 hours [72].
- Optimize Flow Parameters: Use pulsatile flows rather than steady flow, as research indicates optimized frequency and amplitude can significantly delay microchannel clogging compared to steady flow conditions [1].
Problem: Performance Degradation Over Time
Symptoms: Gradual reduction in processing efficiency, increased pressure requirements, decreased sample purity.
Solutions:
- Apply Microbubble Streaming: Implement piezotransducer-activated microbubbles near constrictions to generate 3D counter-rotating vortices that disrupt arch formation and particle clusters. This approach can operate in event-triggered, continuous, or periodic modes to match specific application needs [1].
- Surface Treatment: Modify channel surfaces with anti-fouling coatings or chemical treatments to reduce cell adhesion, though note that this method may have durability limitations for long-term use [72].
- Regular Maintenance Protocol: Establish routine cleaning cycles using appropriate buffers or solutions to prevent cumulative adhesion. The frequency should be determined based on sample type and usage intensity.
Comparative Analysis of Anti-Clogging Technologies
Table 1: Quantitative Comparison of Anti-Clogging Technology Performance
| Technology | Clog-Free Operation Duration | Capture Efficiency | Implementation Complexity | Relative Cost |
|---|---|---|---|---|
| Dielectrophoresis (DEP) | 12+ hours [72] | 97.23% purity efficiency [72] | High (electrodes, power supply) | Medium-High |
| Microbubble Streaming | Limited data (continuous operation demonstrated) [1] | Maintains flow in constrictions [1] | Medium (piezotransducer, controller) | Medium |
| Multifunctional Microposts | No clogging observed in testing [74] | 96% for cancer cells [74] | Low (design-based) | Low |
| Acoustic Focusing | Designed for problematic samples [73] | Compatible with various cell types [73] | High (integrated system) | High |
| Templated Emulsification | Microfluidics-free approach [75] | High-purity transcriptomes [75] | Low (vortexer only) | Low |
Table 2: Cost-Benefit Analysis of Anti-Clogging Approaches
| Technology | Initial Investment | Operational Costs | Maintenance Requirements | Best-Suited Applications |
|---|---|---|---|---|
| Dielectrophoresis | Medium-High (electrode fabrication) | Low (power consumption) | Electrode cleaning, potential fouling | Continuous blood plasma separation [72] |
| Microbubble Streaming | Medium (transducer, controller) | Low (power consumption) | Minimal, bubble replenishment | Lab-on-chip with constrictions [1] |
| Multifunctional Microposts | Low (design-based) | None | Physical integrity checks | High-throughput rare cell isolation [74] |
| Acoustic Focusing | High (commercial system) | Medium (consumables) | Professional servicing | Large or clumpy cells (cardiomyocytes, tumor cells) [73] |
| Templated Emulsification | Low (no specialized equipment) | Low (standard lab equipment) | None | High-scale single-cell genomics [75] |
Experimental Protocols
Protocol 1: Implementing Dielectrophoresis for Blood Plasma Separation
Principle: Apply non-uniform electric fields via integrated electrodes to generate repulsive forces that prevent blood cells from adhering to channel walls [72].
Materials:
- Microfluidic device with interdigitated electrode pairs
- Function generator for AC voltage supply
- Syringe pump with pressure control
- Blood sample with anticoagulant
- Phosphate-buffered saline (PBS)
Procedure:
- Fabricate microfluidic device with electrode pairs at the bottom of branch channels for plasma extraction [72].
- Introduce blood sample into the device using syringe pump at controlled flow rate.
- Apply AC voltage at 20V to electrodes to generate dielectrophoretic forces [72].
- Monitor pressure sensors to confirm stable operation.
- Collect separated plasma from outlet channel for analysis.
- Verify performance by calculating purity efficiency and plasma yield.
Validation:
- Calculate purity efficiency:
(Number of particle-free droplets) / (Total number of droplets) × 100%[72]. - Calculate plasma yield:
(Volume of extracted plasma) / (Theoretical maximum plasma volume) × 100%[72]. - Expected results: 97.23% ± 5.43% purity efficiency and 38.95% ± 9.34% plasma yield at 20V [72].
Protocol 2: Microbubble Streaming for Clog Prevention at Constrictions
Principle: Use acoustically activated microbubbles to generate microstreaming vortices that disrupt particle aggregation near channel constrictions [1].
Materials:
- PDMS microfluidic device with lateral bubble cavities
- Piezoelectric transducer
- Function generator and amplifier
- Fluorescent particles for visualization
- Aqueous solution with glycerol for neutral buoyancy
Procedure:
- Fabricate microfluidic device with lateral cavities (width: 80μm, length: 500μm) near constrictions to trap microbubbles during liquid infusion [1].
- Affix piezoelectric transducer to the microchip.
- Introduce particle suspension into the device.
- Activate transducer at determined resonant frequency (typically 1-10 kHz) to generate microbubble oscillations [1].
- Monitor constriction areas for clog formation using microscopic imaging.
- Operate system in event-triggered, continuous, or periodic mode based on application needs.
Validation:
- Quantify clogging events per unit time with and without microbubble activation.
- Measure particle velocity and distribution patterns near constrictions.
- Expected result: Effective prevention of arch formation and disintegration of existing particle clusters [1].
Technical Diagrams
Clogging Mitigation Decision Pathway
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Anti-Clogging Experiments
| Item | Function | Example Application |
|---|---|---|
| Polydimethylsiloxane (PDMS) | Microfluidic device fabrication | Creating transparent, flexible microchannels for visualization [72] |
| Interdigitated Electrodes | Generating dielectrophoretic forces | Creating repulsive fields in blood plasma separators [72] |
| Piezoelectric Transducers | Activating microbubble oscillation | Generating acoustic microstreaming for clog disruption [1] |
| Polyacrylamide Beads with Poly(T) | mRNA capture in single-cell analysis | Barcoding transcripts in templated emulsification [75] |
| Proteinase K | Temperature-activated cell lysis | Releasing mRNA after compartmentalization in PIP-seq [75] |
| Fluorescent Microspheres | Particle tracking and visualization | Testing device performance and clog formation [1] |
| Surface Treatment Solutions | Reducing cell adhesion | Modifying channel surface properties to prevent fouling [72] |
Selecting appropriate anti-clogging technology requires careful consideration of your specific application requirements, sample types, and operational constraints. Design-based passive methods offer cost-effective solutions for standardized high-throughput applications, while active technologies provide greater flexibility for diverse sample types in research settings. By implementing the protocols and troubleshooting guides provided in this technical support center, researchers can significantly improve the reliability and efficiency of their microfluidic single-cell analysis workflows, ultimately enhancing research productivity and experimental outcomes.
Conclusion
Effective prevention of microfluidic channel blockage requires a multifaceted approach integrating optimized device architecture, strategic platform selection, and rigorous operational protocols. The advancement of active microfluidics, sophisticated surface treatments, and intelligent real-time monitoring represents the future of clog-free single-cell analysis. As these technologies mature, they promise to enhance experimental reproducibility, enable longer culture durations, and facilitate the integration of microfluidics into routine clinical and diagnostic workflows. By adopting the comprehensive strategies outlined across foundational understanding, methodological applications, troubleshooting protocols, and validation frameworks, researchers can significantly improve system reliability and data quality, ultimately accelerating discoveries in fundamental biology and translational medicine.
References
- 1. A method to prevent clogging and clustering in microfluidic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11232117/]
- 2. Mitigating clogging in a microfluidic array [https://www.elveflow.com/microfluidics-research-summaries/microfluidic-arrays-clogging-mitigation/]
- 3. A simple trick to open up clogged microfluidic chip [https://blogs.rsc.org/chipsandtips/2013/07/18/a-simple-trick-to-open-up-clogged-microfluidic-chip/]
- 4. Affordable method for channel geometry–specific flow ... [https://www.nature.com/articles/s41598-025-24442-5]
- 5. Effect of surface energy and roughness on cell adhesion ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8698786/]
- 6. Microfluidic Single-Cell Manipulation and Analysis [https://www.mdpi.com/2072-666X/10/2/104]
- 7. Troubleshooting Protocols - Biological Sciences: Summer ... [https://guides.library.cmu.edu/biologicalsciencesSRI/troubleshooting]
- 8. Sampling time-dependent artifacts in single-cell genomics ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7212672/]
- 9. Clogging-free microfluidics for continuous size-based ... [https://www.nature.com/articles/srep26531]
- 10. Empowering single-cell analysis with emerging active ... [https://www.sciencedirect.com/science/article/abs/pii/S0142961225005368]
- 11. Microfluidic Platforms for Ex Vivo and In Vivo Gene Therapy [https://pmc.ncbi.nlm.nih.gov/articles/PMC12384408/]
- 12. Advances in Microfluidic Single-Cell RNA Sequencing and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12029715/]
- 13. Calculating the cost of downtime [https://www.atlassian.com/incident-management/kpis/cost-of-downtime]
- 14. The True Cost of IT Downtime [https://techmate.com/blog/the-true-cost-of-it-downtime/]
- 15. Business Impact Analysis [https://www.ready.gov/business/planning/impact-analysis]
- 16. Prepare for Impact with a Business Impact Analysis [https://frsecure.com/blog/business-impact-analysis/]
- 17. Troubleshooting Basics, Part II: Pressure Problems [https://www.chromatographyonline.com/view/troubleshooting-basics-part-ii-pressure-problems]
- 18. Enhancing single-cell encapsulation in droplet ... [https://www.nature.com/articles/s41378-023-00631-y]
- 19. Enhanced single-cell encapsulation in microfluidic devices [https://pmc.ncbi.nlm.nih.gov/articles/PMC7758092/]
- 20. A microfluidic device enabling high-efficiency single cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4288539/]
- 21. Best Surfactants for Microfluidic Droplet Generation [https://blog.darwin-microfluidics.com/best-surfactants-microfluidic-droplet-generation/]
- 22. Droplet-Sequencing (Drop-Seq) [https://elveflow.com/microfluidic-reviews/drop-seq/]
- 23. These pneumatic troubleshooting charts cover many ... [https://www.valmet.com/insights/articles/up-and-running/reliability/RTPneuTrouble/]
- 24. What Techniques Reducing Pneumatic Actuator Speed [https://www.valen-tech.com/what-techniques-reducing-pneumatic-actuator-speed.html]
- 25. How to Troubleshoot Common Issues in Pneumatic Systems [https://pennair.com/2024/09/06/how-to-troubleshoot-common-issues-in-pneumatic-systems/]
- 26. Hydrodynamic Cell Trapping for High Throughput Single- ... [https://www.mdpi.com/2072-666X/4/4/414]
- 27. Microfluidic systems for hydrodynamic trapping of cells and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8719525/]
- 28. Membrane Fouling Phenomena in Microfluidic Systems [https://pmc.ncbi.nlm.nih.gov/articles/PMC8305447/]
- 29. Design and Application of Antifouling Bio-Coatings - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11945268/]
- 30. Progress in Organic Coatings [https://www.sciencedirect.com/science/article/abs/pii/S0300944023006665]
- 31. Typical Failure Modes In Microfluidics [https://www.alineinc.com/typical-failure-modes-in-microfluidics/]
- 32. Filtration Techniques in Sample Preparation [https://www.phenomenex.com/knowledge-center/spe-knowledge-center/filtration-techniques?srsltid=AfmBOopvJ_9Q_j4IY-36d3L_G2WR17x03NxAGFwB2TLCHcdjUrQExEkk]
- 33. Optimization Techniques: General Guidelines [https://www.worthington-biochem.com/tools-resources/tissue-dissociation-guide/optimization-techniques-general-guidelines]
- 34. InTraSeq™ Single Cell Analysis Frequently Asked Questions [https://www.cellsignal.com/applications/intraseq-single-cell-faq?srsltid=AfmBOoq1IAtDAYc-6OhBx4lwbEGAlqRJ2PPtcacFKfM1p_YkhwQr5i6C]
- 35. Sample Preparation by Filtration [https://www.sigmaaldrich.com/US/en/applications/analytical-chemistry/sample-preparation/filtration?srsltid=AfmBOoo5y-gBJxHn8WkDVLdz8d8Gnu-QJMsz6VFasSnBklMT3WX5ksDL]
- 36. 8 Ways to Optimize Cell Cultures [https://vistalab.com/8-ways-to-optimize-cell-cultures/]
- 37. Understanding and Managing Pressure in Microfluidic ... [https://elveflow.com/microfluidic-applications/pressure-sensor-feedback-loop/]
- 38. Flow Cytometry Support—Troubleshooting [https://www.thermofisher.com/us/en/home/technical-resources/technical-reference-library/cell-analysis-support-center/flow-cytometry-support/flow-cytometry-support-troubleshooting.html]
- 39. Data Diagnostics Theory - Upstream Oil & Gas [https://www.ihsenergy.ca/support/documentation_ca/Harmony/content/html_files/reference_material/general_concepts/data_diagnostics_theory.htm]
- 40. How to Perform a Microfluidic Cultivation Experiment—A ... [https://www.mdpi.com/2079-6374/11/12/485]
- 41. The Practical Guide to Cleaning Microfluidic Devices [https://www.thierry-corp.com/plasma-treatment-articles/the-practical-guide-to-cleaning-microfluidic-devices]
- 42. Cell culture and maintenance protocol [https://www.abcam.com/en-us/technical-resources/protocols/cell-culture-and-maintenance-protocol]
- 43. Recent advances in microfluidic platforms for single-cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7434086/]
- 44. How to Perform a Microfluidic Cultivation Experiment—A ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8699335/]
- 45. Microfluidic chip for precise trapping of single cells and ... [https://www.nature.com/articles/s44172-022-00019-2]
- 46. Preventing Bubble Entrapment in Microfluidic Devices ... [https://www.comsol.it/blogs/preventing-bubble-entrapment-in-microfluidic-devices-using-simulation]
- 47. FAQs about single cell sample preparation (covering the ... [https://www.10xgenomics.com/blog/faqs-about-single-cell-sample-preparation-covering-the-basics]
- 48. Cell Preparation Considerations and Troubleshooting [https://www.aatbio.com/resources/application-notes/cell-preparation-considerations-and-troubleshooting]
- 49. How to Separate Dead Cells and Cellular Debris [https://www.akadeum.com/types-of-cell-separation/dead-cell-removal/?srsltid=AfmBOor0ZaQTQJYg7VwCB7TydpZiMqi6E_QJiJfsk9NHcaIKuZ637045]
- 50. 7 Things to Consider When Preparing a Cell Suspension ... [https://www.parsebiosciences.com/blog/cell-prep-for-single-cell-sequenci/]
- 51. Best Practices for Preparing a Single Cell Suspension from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6375754/]
- 52. Sterilization guide for microfluidic medical devices [https://www.parallelfluidics.com/resources/knowledge-base/sterilization-guide-for-microfluidic-medical-devices]
- 53. Safety & Protocols | Harvard Medical School [https://hms.harvard.edu/departments/hms-microfluidics/microfabrication-facility/safety-protocols]
- 54. Hydro-Seq enables contamination-free high-throughput ... [https://www.nature.com/articles/s41467-019-10122-2]
- 55. What Influences the Safety and Compliance of Microfluidic Chips? [https://eureka.patsnap.com/report-what-influences-the-safety-and-compliance-of-microfluidic-chips]
- 56. A brief overview of passive microvalves in microfluidics [https://pmc.ncbi.nlm.nih.gov/articles/PMC11037934/]
- 57. Microfluidics FAQ and Product Information | AMF [https://amf.ch/faq/]
- 58. Long-Term Channel Block Is Required to Inhibit Cellular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4127929/]
- 59. Overcoming technological barriers in microfluidics [https://pmc.ncbi.nlm.nih.gov/articles/PMC9490024/]
- 60. Measurement of Microchannel Fluidic Resistance with a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3549673/]
- 61. Innovative Advances in Droplet Microfluidics - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12384975/]
- 62. Droplet Microfluidics for Advanced Single‐Cell Analysis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11970111/]
- 63. A design and optimization of a high throughput valve ... [https://www.nature.com/articles/s41598-021-92472-w]
- 64. spinDrop: a droplet microfluidic platform to maximise single ... [https://www.nature.com/articles/s41467-023-40322-w]
- 65. Recent advances in droplet microfluidics for single-cell ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993623000195]
- 66. Emerging Trends in Microfluidics [https://elveflow.com/microfluidic-reviews/emerging-trends-in-microfluidics/]
- 67. Microfluidic Sample Preparation for Single Cell Analysis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4809371/]
- 68. Microfluidic Chips for Single-cell Analysis and Isolation [https://www.ufluidix.com/microfluidics-applications/single-cell-analysis/]
- 69. Microfluidics applications for high-throughput single cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8507141/]
- 70. IHC Troubleshooting Guide [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/ihc-troubleshooting-guide.html]
- 71. Troubleshooting Guide: Immunohistochemistry [https://www.rndsystems.com/resources/technical/troubleshooting-guide-immunohistochemistry]
- 72. An anti-clogging method for improving the performance ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6242854/]
- 73. Anti-Clogging Technology of the Attune NxT Flow Cytometer [https://www.thermofisher.com/jp/en/home/life-science/cell-analysis/flow-cytometry/flow-cytometers/attune-acoustic-focusing-flow-cytometer/anti-clogging-technology-attune-nxt-flow-cytometer.html]
- 74. Clog-free high-throughput microfluidic cell isolation with ... [https://www.nature.com/articles/s41598-021-94123-6]
- 75. Microfluidics-free single-cell genomics with templated ... [https://www.nature.com/articles/s41587-023-01685-z]